Abstract
Three cats with upper respiratory tract obstruction due to laryngeal inflammation are presented. Cervical radiography showed the presence of a soft tissue mass in the laryngeal region in all cases, and laryngoscopy allowed direct visualization of a mass associated with the larynx. Laryngeal samples were obtained by a combination of fine needle aspiration, cutting biopsy forceps, by ventral laryngotomy, and at post-mortem. Histopathology of the laryngeal samples showed the presence of a predominantly granulomatous inflammation, with macrophage and lymphocyte infiltration. One case was euthanased due to severe dyspnoea. The remaining two cases underwent combined medical (corticosteroid and antibiotic) and surgical (permanent tracheostomy or excision of laryngeal tissue by ventral laryngotomy) treatment. One case died of an undetermined cause 15 weeks after surgery while the other case remains clinically well 20 months after diagnosis. Recognition of the existence of granulomatous laryngitis is important as clinical signs and radiographic findings are indistinguishable from laryngeal neoplasia.
Inflammatory laryngeal disease in the dog and cat tends to be acute and rarely results in life-threatening respiratory obstruction. Examples include laryngeal inflammation due to infectious agents or following foreign body penetration, and oedema secondary to insect bites (Harvey & O'Brien 1982, Venker-van Haagen 1992, Holt & Brockman 1994). In dogs chronic laryngeal inflammation is relatively common (Venker-van Haagen 1992) but resulting respiratory obstruction is rare (Oakes & McCarthy 1994, Harvey & O'Brien 1982, Wheeldon et al 1982). A review of the literature revealed only three cases of laryngeal inflammation which resulted in respiratory obstruction in cats (Harvey & O'Brien 1982). These cats were included in a case series on the surgical treatment of miscellaneous laryngeal diseases, but detailed descriptions of the clinical presentation, radiological findings and laryngeal histopathology were not given, and specific treatment was not described. The major reported causes of upper respiratory distress in cats include pharyngeal polyps, laryngospasm, laryngeal or pharyngeal trauma, tracheal or laryngeal foreign body penetration, and neoplasia of the trachea or larynx (Turnwald 1995). This paper presents the historical, clinical, clinicopathological, radiological and histopathological findings in three cats with upper respiratory tract obstruction due to granulomatous laryngitis (GL), and describes treatments and outcomes.
Materials and Methods
The case records were reviewed retrospectively. The three cats were presented to the Royal (Dick) School of Veterinary Studies at The University of Edinburgh for investigation of dyspnoea. A thorough history was obtained and clinical examination performed in each case. In all cats blood samples were obtained for routine haematology and serum biochemistry screens, feline leukaemia virus (FeLV) antigen testing using an enzyme-linked immunosorbent assay (ELISA) test (Inochem, C. Lutz), and feline immunodeficiency virus (FIV) antibody test using immunofluorescence. Oropharyngeal swabs for feline herpesvirus and calicivirus isolation were collected from one cat.
Thoracic and lateral cervical radiographs were taken under sedation [0.1 mg/kg intramuscular acetylpromazine, (ACP; C-Vet) alone or in combination with 5 mg/kg intramuscular ketamine (Vetalar; Pharmacia & Upjohn Ltd)]. Under light propofol (Rapinovet; Schering-Plough Animal Health) anaesthesia, and before intubation, the larynx was examined by direct per os laryngoscopy and endoscopy (BF-30, Keymed). General anaesthesia was induced in all cases with 6 mg/kg intravenous propofol and maintained with halothane and oxygen once intubation was possible. Samples of laryngeal tissue were collected by a combination of fine needle aspiration (FNA) per os, cutting biopsy forceps per os and/or by ventral laryngotomy. Additional laryngeal samples were collected at post-mortem examination in one cat. These samples were submitted for cytological or histopathological examination. Special stains (Ziehl-Neelsen, Periodic acid-Schiff and Gram) were used on laryngeal biopsy samples from cases 1 and 2.
Results
Signalment and history
The cats were aged 7, 9 and 12 years. Dyspnoea over a period of 10 days to 5 months prior to presentation was reported by the owners (Table 1). Cases 1 and 2 had chronic intermittent but progressively worsening clinical signs, while case 3 presented with a sudden onset of relatively severe clinical signs. Inspiratory dyspnoea and coughing were reported in all cats. Other clinical signs included lethargy, dysphonia, inappetance and retching. All three cats had received treatment by the referring veterinarian prior to assessment, with combinations of steroids, non-steroidal anti-inflammatory agents and antibiotics. However, only slight and temporary improvement in clinical signs was seen.
Case details and presenting clinical signs

FN; female neutered. MN; male neutered.
Clinical examination
Mild to marked inspiratory effort was apparent in all cats, with stridor being audible in cases 2 and 3. Respiratory rates varied between 20 and 40 breaths per minute. Auscultation of the chest in all cases revealed increased respiratory sounds (rhonchi) which were referred from the upper respiratory tract and were loudest over the laryngeal area. Palpation of the larynx was considered normal. Cases 2 and 3 had grade II/VI systolic heart murmurs audible on cardiac auscultation but heart rates and rhythm were normal.
Clinicopathological examination
Haematology revealed a mild eosinophilia (1.224×10/l; normal range 0.0–1.0×109/l) in case 1 and a moderate mature neutrophilia (35.52×109/l; normal range 2.5–12.8×109/l) in case 3. A moderately severe hypoalbuminaemia (15.5 g/l; normal range 28–35 g/l) in case 3 was not investigated further. There were no other biochemical abnormalities. All virus tests were negative.
Radiological assessment
Thoracic radiography in case 1 was unremarkable. Case 2 had evidence of aerophagia (air within the oesophagus and stomach). Case 3 had a mild bronchial pattern throughout the lung fields. In all cats, a soft tissue density almost totally occluding the laryngeal lumen was visible on cervical radiography (Fig 1).
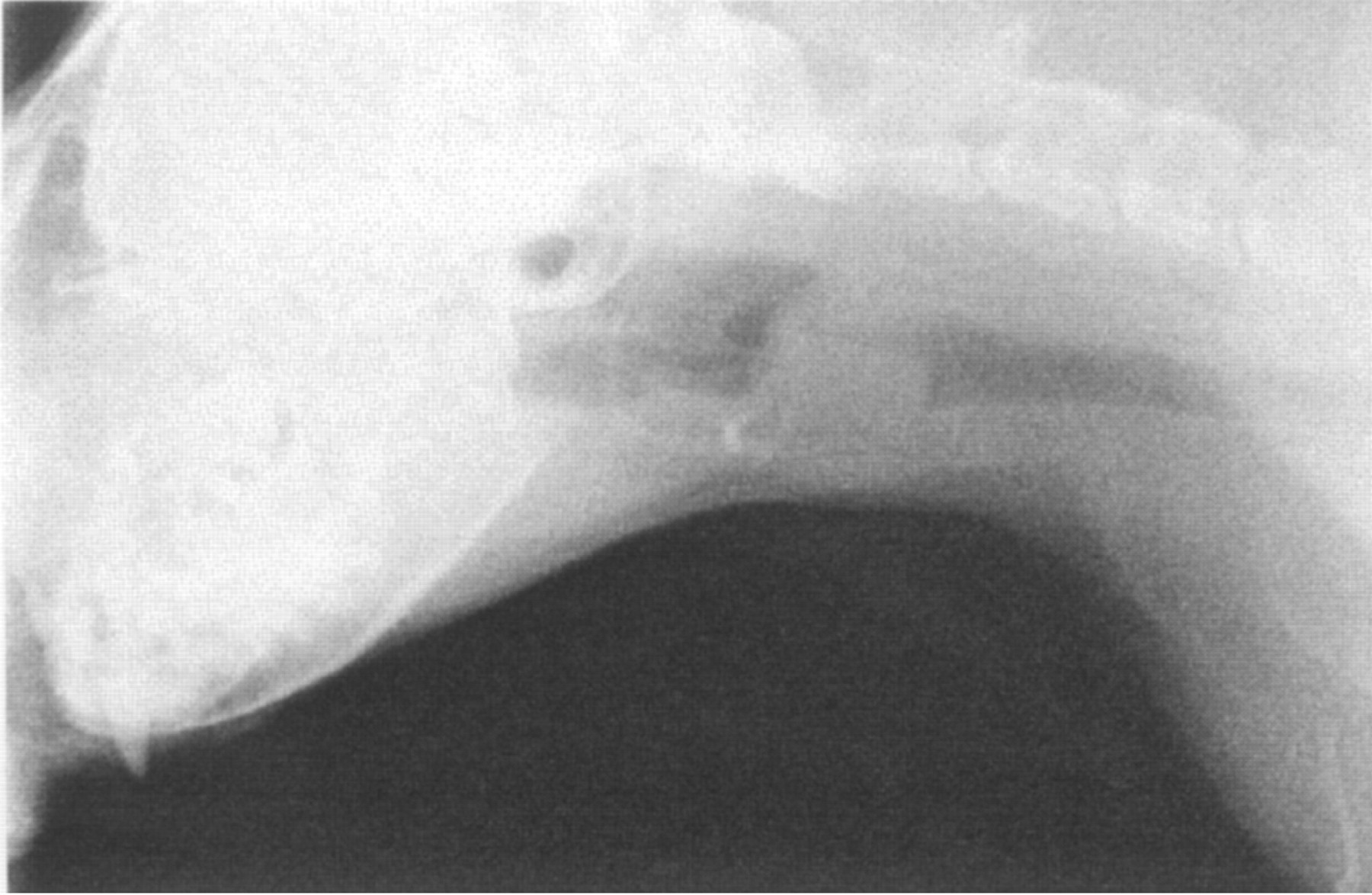
Lateral cervical radiograph from case 2 illustrating the typical radiographic appearance. There is an overall increase in soft tissue density obscuring the anatomical detail of the larynx.
Laryngoscopy findings
Laryngoscopy findings are summarized in Table 2. In all cats, a soft tissue mass, almost totally occluding the rima glottis, was apparent (Fig 2a). The mass appeared to originate from the left side in cases 1 and 3, and was bilateral in case 2.
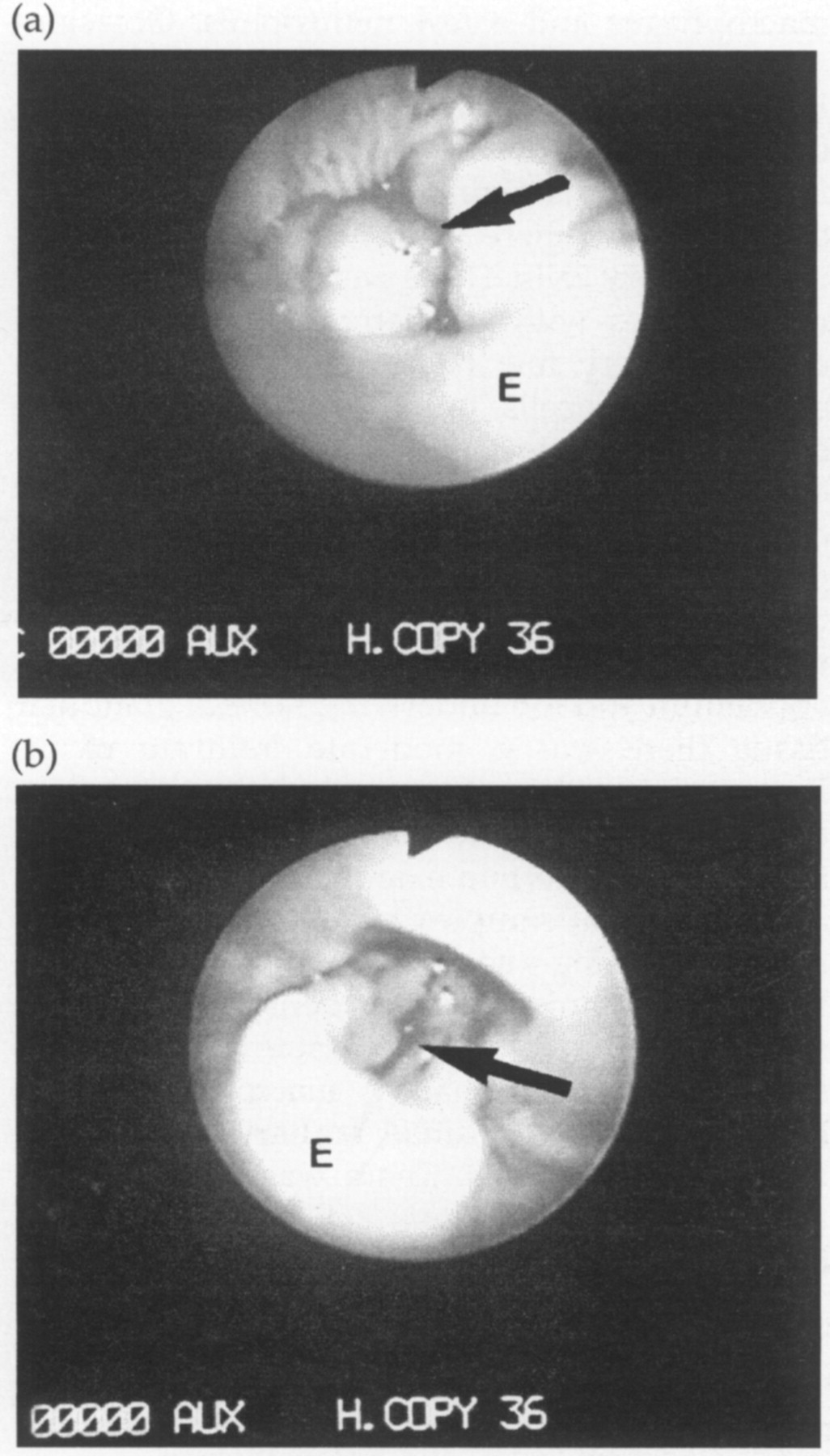
Endoscopic views of the larynx in case 2. (a) View of the larynx at initial presentation. There are severe proliferative changes resulting in near total occlusion of the rima glottis. (b) View of the larynx after 5 days of glucocorticoid treatment. There is marked improvement in the appearance of the larynx, with a reduction in proliferative tissue, although the rima glottis is still significantly occluded. The remaining proliferative changes appear to be associated with the arytenoids.; Key: E, epiglottis; Arrow, glottis.
Summary of laryngeal biopsy methods and histopathological findings

FNA: fine needle aspirate.
Histopathological results
Histopathological results are summarized in Table 2.
In Case 1 cytological examination of a FNA sample revealed inflammatory changes with a predominance of lymphocytes, low numbers of macrophages and a few neutrophils. Occasionally bacteria were present within the neutrophils. Laryngeal tissue obtained at post-mortem examination (7 days later; Fig 3) revealed an intact non-keratinizing squamous epithelium beneath which there was an intense infiltrate of inflammatory cells. These were lymphocytes and macrophages with smaller numbers of plasma cells, but very few neutrophils. There was no exocytosis of cells onto the surface and the connective tissue immediately beneath the epithelium showed only mild inflammatory changes. The laryngeal biopsy covered by pseudo-stratified respiratory epithelium (and therefore taken from the distal part of the larynx) was substantially ulcerated and between this epithelium and the underlying normal glandular tissue there was a moderate infiltrate of inflammatory cells, which included neutrophils, and some oedema. No other abnormalities were found at post mortem examination.
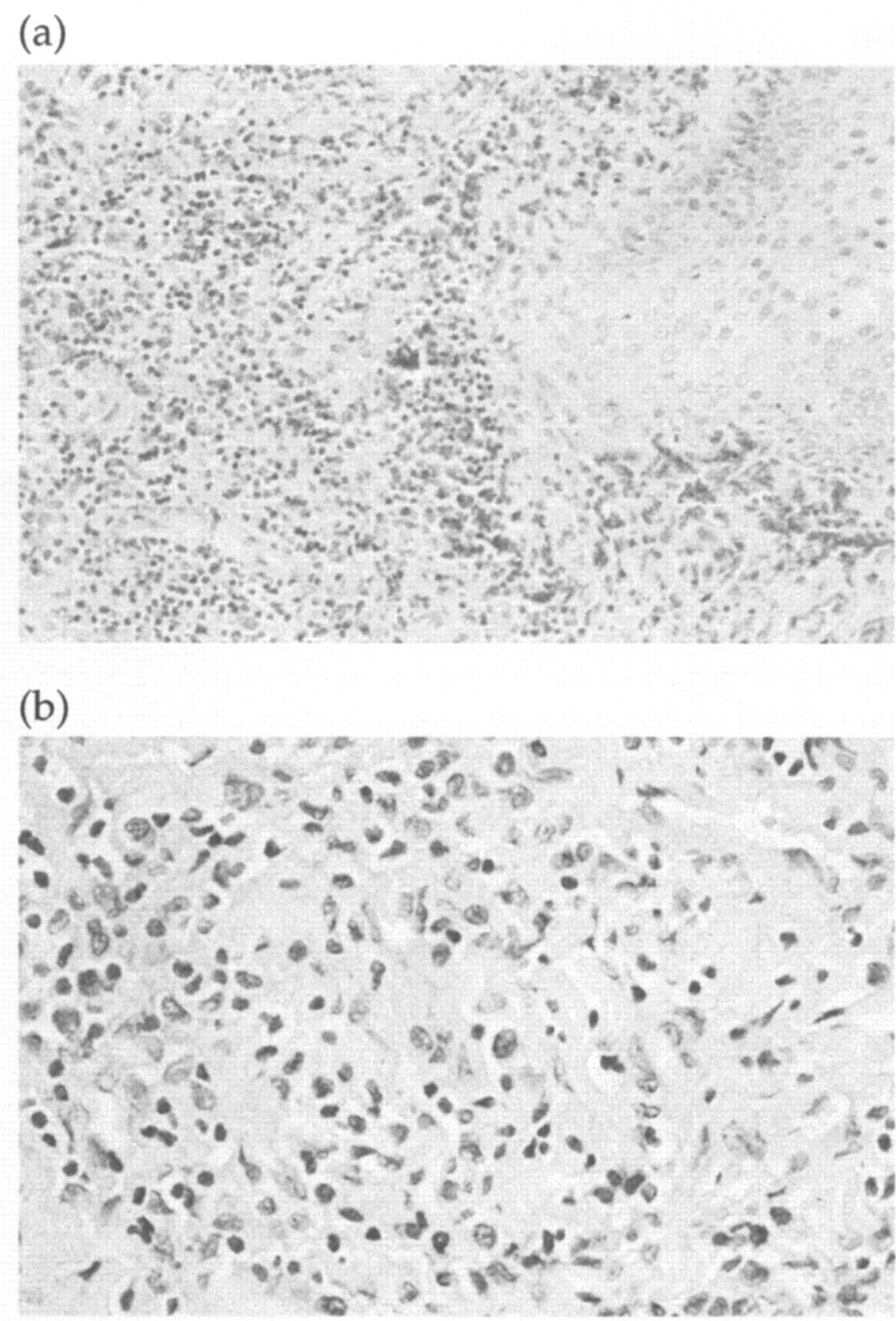
Histopathology of laryngeal tissue from case 1 (H&E) (a) Squamous epithelium is visible to the right (some crush artefact is present dorsally). A marked cellular infiltrate is present within the tissue to the left of the epithelium. (×100). (b) High power magnification of the cellular infiltration in (a) (×400). Lymphocytes and macrophages are the predominant cell types in this granulomatous inflammation.
All laryngeal samples taken from case 2 had non-keratinizing squamous epithelium. The first biopsy showed a very dense cell infiltrate underlying the connective tissue (Fig 4). These cells were predominantly macrophages with lymphocytes, and a small scattering of plasma cells and neutrophils. There was no exocytosis through the squamous epithelium and a narrow band of connective tissue with very mild inflammation was present between the squamous epithelium and the inflammatory mass. Within this superficial area there was also marked dilatation of lymphatic vessels. No ulceration was present. The second biopsy sample obtained 5 days later revealed similar deep inflammation with macrophages and lymphocytes, but, in addition, an area of more acute inflammation was present, where the epithelium had ulcerated.

Histopathology of laryngeal tissue from case 2 (H&E) (a) L indicates a band of mature lymphocytes above which is a large area of granulomatous tissue (G). (×100) (b) High power magnification of the granulomatous tissue in (a) (×400). Macrophages are the predominant cell type with some lymphocytes.
The first laryngeal biopsies from case 3 had intact stratified squamous epithelium overlying an area of granulation tissue with chronic inflammation, comprising macrophages and lymphocytes. An excisional biopsy 5 days later had similar granulomatous inflammation, with mainly macrophages and lymphocytes, and occasional neutrophils.
Special stains failed to identify organisms on the larygneal biopsies taken from cases 1 and 2.
The histopathological diagnosis in all cases was granulomatous laryngitis.
Outcome
Case 1 was euthanased at the owner's request due to severe dyspnoea while cases 2 and 3 were treated medically and surgically.
Case 2 had a temporary tube tracheotomy performed at the time of the first laryngeal biopsy, because of severe inspiratory dyspnoea. Post-operatively the cat was given intravenous dexamethasone (Soludex; Intervet UK Ltd) at a dosage of 0.2 mg/kg/day and oral amoxycillin with clavulanic acid (Synulox; Pfizer Ltd) at 30 mg/kg/day whilst awaiting results. Five days later the cat was re-anaesthetized due to ongoing inspiratory dyspnoea. Laryngoscopy showed some reduction in the size of the laryngeal mass (Fig 2b) but the laryngeal opening was still considered severely compromised. The obstruction was bypassed by permanent tracheostomy and a repeat laryngeal biopsy taken per os. Dexamethasone was continued post-operatively for 2 days and the cat continued on oral prednisolone (Prednicare; Animalcare Ltd) (initially 1 mg/kg/day, reducing after 2 weeks) and amoxycillin with clavulanic acid for 2 weeks. The cat was asymptomatic for 4 weeks before developing a progressively worsening cough. Nine weeks after permanent tracheostomy the laryngeal mass was still visible on endoscopy, although smaller, and on thoracic radiography, compression of the trachea at the carina was noted. Tracheobronchoscopy confirmed partial compression of the trachea at the carina with roughened mucosa. Endoscopic punch biopsies of the mucosa demonstrated lymphoplasmacytic inflammation. Corticosteroid therapy was re-instituted with 0.3 mg/kg/day oral dexamethasone (Opticorten; Novartis Animal Health UK Ltd) and the coughing resolved. The cat remained asymptomatic, with no signs of respiratory distress, on low dose (0.07 mg/kg/day) dexamethasone but died suddenly 15 weeks after permanent tracheostomy. A post-mortem examination was not permitted.
Case 3 had a ventral laryngotomy and temporary tracheotomy performed for excision of the laryngeal mass. Post-operatively the cat was given 20 mg/kg intravenous methylprednisolone (Solu-Medrone V; Pharmacia & Upjohn Ltd) and 20 mg/kg intravenous cephalexin (Kefzol; Eli Lilly & Co Ltd) and started on a two week course of oral amoxycillin with clavulanic acid (30 mg/kg/day) with oral prednisolone (initially 3 mg/kg/day). The temporary tracheotomy tube was removed on post-operative day 2 and the cat was discharged on continuing treatment. Prednisolone treatment was tapered down over a period of 6 months and then stopped. Twenty months following surgery the cat was well and was not on any treatment.
Discussion
Upper airway obstruction due to inflammatory laryngeal disease appears to be rare in the cat, but should be considered in the differential diagnoses for feline dyspnoea. This paper describes three cats with GL. All cats presented with signs of upper respiratory tract obstruction. Three feline cases with upper respiratory tract obstruction due to laryngeal inflammation were previously reported in a series describing surgical treatment of miscellaneous laryngeal conditions in the dog and cat (Harvey & O'Brien 1982). A diagnosis of inflammatory laryngitis was obtained by histopathology but detailed histopathological descriptions from these cases were not given. Two of the cats were described as having chronic proliferative, ulcerative or granulomatous laryngitis and had an asymmetrical lesion visible by laryngoscopy. The other cat in that series, whose clinical signs had been acute and sudden in onset, was described as having non-granulomatous laryngeal inflammation. The clinical signs seen in these three cats included respiratory distress, retching and coughing.
The pathological changes seen in all the cases of the current report were similar. They were chronic and granulomatous in type, involving macrophages, lymphocytes and plasma cells, with little neutrophil involvement. All cases had densely packed nodular granulomatous lesions within the area of the larynx covered by non-keratinizing squamous epithelium. Additional laryngeal biopsies from case 1 were covered by pseudo-stratified respiratory epithelium, and also showed a moderate inflammatory infiltrate, including some neutrophils, but the inflammation was not as severe as the granulomatous type seen in squamous area of the larynx. Neutrophils were present in some laryngeal samples, suggesting more acute inflammatory changes. This was found to be a feature in those biopsies taken after a previous laryngeal sampling procedure had been performed, and may have been a response to the injury induced by sampling of the laryngeal tissue.
GL is a rare but well recognized disease in humans and results in hoarseness, dyspnoea and coughing (Brass & White 1991, McFerran et al 1994). Histopathological findings on laryngeal biopsies are similar to those presented in the cats in our study and consist of a mixed inflammatory cell infiltrate involving macrophages, lymphocytes and plasma cells. Ulceration of the epithelium is a common finding in humans, and was present in some areas of the biopsies taken from two of the three cases in the current report. In humans, GL can be due to specific disease processes such as tuberculosis, syphilis and fungal infections, but non-specific GL is usually secondary to chronic abuse of the vocal cords or intubation. An idiopathic form of GL is also recognized (McFerran et al 1994) where no underlying cause is discovered.
No specific aetiology for the GL in the cats in our study was found. Special stains did not identify organisms on laryngeal biopsies in two cases and, although the presence of bacteria on the FNA in case 1 might suggest an infectious cause, mixed bacterial populations are considered normal oropharyngeal flora. A mild secondary bacterial infection could have arisen secondary to the ulceration. No evidence of foreign material was seen on histopathology in any case. In a report by Wheeldon et al (1982) trauma was suspected to be the cause of chronic GL in two dogs. An interesting feature seen on histopathology of the laryngeal biopsies in two of our cases was the presence of a narrow zone of less inflammed tissue immediately beneath the epithelium. This suggested that the inflammation was developing deep within the tissue rather than extending from the surface, making foreign body penetration or trauma less likely to be causal.
Although GL in the cat is rare, recognition of the existence of such inflammatory conditions is important. In the three cats presented in this series, the laryngeal disease was clinically and radiographically considered most likely to be neoplastic and this contributed to the decision to euthanase one of the cats. However, primary laryngeal tumours are rare in the dog and cat (Saik et al 1986, Carlisle et al 1991). Lymphosarcoma and squamous cell carcinoma are the most commonly reported laryngeal tumours in cats (Venker-van Haagen 1992). Feline laryngeal neoplasia has been reported in a wide age range (Saik et al 1986), similar to that of cats with inflammatory conditions, and both laryngeal neoplasia and laryngeal inflammation cause clinical signs of upper airway disease and radiographic changes that are indistinguishable (Wheeldon et al 1982). The gross appearance of laryngitis can also be very similar to laryngeal neoplasia, so histopathological examination of laryngeal tissues is essential to obtain the correct diagnosis (Harvey & O'Brien 1982).
Combined medical and surgical treatment of laryngeal inflammation causing respiratory obstruction in the dog and cat has been reported (Harvey & O'Brien 1982, Oakes & McCarthy 1994), although specific treatment of the feline cases was not described. Surgical excision of the proliferative laryngeal tissue by partial laryngectomy has been reported, but intermittent or long-term prednisolone was required in some cases to control clinical signs. All three cats in our series had corticosteroid treatment prior to referral, with only slight and temporary improvement in clinical signs. Case 1 had also received prolonged antibiotic treatment prior to referral with partial response. In case 2, a definite improvement in the degree of laryngeal obstruction was observed following high dose intravenous steroid and antibiotic treatment (Fig 2), but this was not sufficient to avoid the need for permanent tracheostomy. In cases 2 and 3 post-operative 2-week courses of antibiotics were given, because the aetiology of the GL had not been confirmed, but further medical treatment comprised of corticosteroids only. Since no evidence for bacterial infection was found in any of the biopsies in this report, the need for antibiotic treatment in GL is questionable. From the limited data in this series we would suggest that surgical treatment, additional to medical treatment with corticosteroids, is required to obtain a favourable outcome.
A good long term prognosis for proliferative inflammatory laryngitis has been reported by some authors (Harvey & O'Brien 1982, Oakes & McCarthy 1994). In the current series specifically describing GL, Case 1 was euthanased before a definitive diagnosis was obtained and case 2 died suddenly of undetermined causes 15 weeks after permanent tracheostomy. The tracheal mucosa at the carina was found to be compressed and roughened on re-examination of this cat, suggesting that a diffuse inflammatory or neoplastic disease was present. It is possible that in this case underlying neoplasia was present, resulting in tracheobronchial lymph node enlargement, but no evidence of neoplasia was seen in any of the biopsy samples taken from this cat. Case 3 remains alive and well 20 months after diagnosis.
In conclusion, GL should be considered as a differential diagnosis in cats which present with upper respiratory tract obstruction due to a laryngeal mass. Confirmation of the diagnosis relies upon histopathological examination of laryngeal tissue. Appropriate surgical and medical management of GL resulted in a long-term favourable outcome in one of the three cases.
Footnotes
Acknowledgements
S Tasker and D J Foster were funded by the Feline Advisory Bureau, whilst these cases were attended. We wish to thank C Belford of Cytopath who performed the cytological examination of the fine needle aspirate, the Department of Veterinary Pathology at the Royal (Dick) School of Veterinary Studies for histopathological examination of biopsies from case 3, and L Genovese of Abbey Veterinary Services, Newton Abbot for initial histopathological examination on cases 1 and 2.
