Abstract

Epidemiologic evidence has now been published in the United States showing a strong association between the administration of inactivated feline vaccines [feline leukaemia virus (FeLV) and rabies] and subsequent soft tissue sarcoma development at sites where these vaccines have been administered (Dubielzig et al 1993, Hendrick & Goldschmidt 1991, Hendrick et al 1992, 1994, Kass et al 1993). The prevalence of soft tissue sarcoma development at sites of vaccination has been reported as 3.6 cases/10 000 to 1 case/10 000 FeLV or rabies vaccines administered (Kass et al 1993, Hendrick et al 1994, Coyne et al 1997). Some believe that the prevalence may be as high as 1/1000 FeLV or rabies vaccines administered (Macy & Hendrick 1996). If these prevalence rates are to be applied to the 1991 United States cat population, the following projections of the number of vaccine-associated sarcomas that occurred that year can be made. In 1991, the United States' cat population was estimated at 57 million cats (Macy & Hendrick 1996). Approximately 62% of cats see veterinarians during any given year, and 64% of the visits to veterinarians include vaccination. These data indicate that 22 million cats were vaccinated in 1991 (Macy & Hendrick 1996). Applying the vaccine-induced tumour prevalence rate of one tumour per 10 000 vaccines administered, approximately 2200 cases of vaccine-associated sarcomas occurred in 1991. Using the higher estimated vaccine-associated sarcoma prevalence of 1/1000 vaccinations, a total of 22 000 vaccine-associated sarcomas occurred in 1991. Because of the relatively low incidence of fibrosarcomas in cats in general (20/100 000), the association between vaccination and subsequent tumour development was made only after millions of doses of now-incriminated vaccines (rabies and FeLV) had been given to cats for half a decade. The number of vaccine-associated sarcomas submitted to veterinary diagnostic laboratories appears to have stabilized and has not increased during the past few years (personal communication).
The first report proposing an association between vaccination and subsequent fibrosarcoma development in cats was a letter to the editor published in the Journal of the American Veterinary Medical Association in 1991 (Hendrick & Goldschmidt 1991). Other reports followed, some citing increased numbers of fibrosarcomas diagnosed at diagnostic laboratories ranging between 21 and 61% in cats predominantly at sites routinely used for vaccination (Dubielzig 1993, Hendrick et al 1992, 1994, Kass et al 1993). The largest increases were reported in diagnostic laboratories on the East Coast where there has been a rabies epidemic in wildlife for almost two decades (Hendrick et al 1992).
The national increased prevalence of fibrosarcomas in cats paralleled the introduction and widespread use of two killed adjuvanted vaccines not previously used in the cat in the United States. In 1985, a subcutaneously administered killed aluminium-adjuvanted rabies vaccine was approved for use in cats, and largely replaced the use of the intramuscularly administered high-egg passage modified-live virus rabies vaccine in the United States. [Imrab 3 Rabies Vaccine killed virus, Rhone Merieux] In the same year, for the first time, a killed aluminium-adjuvanted feline leukaemia virus vaccine was also introduced to the veterinary profession (personal communication). Both vaccines received wide acceptance and have been used by veterinarians since their introduction to immunize cats against rabies and FeLV in the United States. The fact that the prevalence of vaccine-associated sarcomas appears to be stabilized may be attributed to the fact that cats entering the annual revaccination pipeline in 1985 have reached their life expectancy, and the prevalence of vaccine-associated sarcomas should remain steady unless there is a change in FeLV and/or rabies vaccines or vaccination protocols for cats. Vaccine-site associated sarcomas are believed to develop in areas of inflammation produced by these adjuvanted vaccine products, although they have also been reported with non-adjuvanted feline vaccines, albeit less often (Hendrick et al 1994, Kass et al 1993, Esplin et al 1993, Fawcett & Smith 1984, Hendrick & Dunagan 1991, Lester et al 1996). Microscopically, areas of transition between inflammation and tumour development have been frequently observed in vaccine-associated sarcomas (Hendrick et al 1992, Esplin et al 1993). The neoplasms that develop after vaccination are typically mesenchymal in origin; fibrosarcomas, malignant fibrous histiocytomas (also referred to as myofibroblastic sarcomas), osteosarcomas, chondrosarcomas, undifferentiated sarcomas, and rhabdomyosarcomas are most frequently reported (Dubielzig et al 1993, Hendrick & Brooks 1994). Vaccine-site sarcomas are histologically similar to mesenchymal tumours that arise in traumatized eyes of cats, suggesting a common pathogenesis of inflammation and wound healing in the development of tumours in these two syndromes (Dubielzig 1984, Dubielzig et al 1990, Woog et al 1983). The presence of inflammatory cells, fibroblasts and myofibroblasts in and around vaccine site sarcomas supports this hypothesis (Fig 1).

Vaccine-associated sarcoma. Note the presence of inflammation (arrow) adjacent to neoplastic tissue (400×).
A comparison of the morphologic features of vaccine site and non-vaccine site sarcomas found significant differences between the two groups of tumours (Doddy et al 1996). Vaccine site sarcomas typically have increased amounts of necrosis, inflammatory cells (mostly lymphocytes and macrophages), and increased numbers of cycling cells as determined by the presence of mitosis when compared with non-vaccine site sarcomas (Fig 2) (Doddy et al 1996).
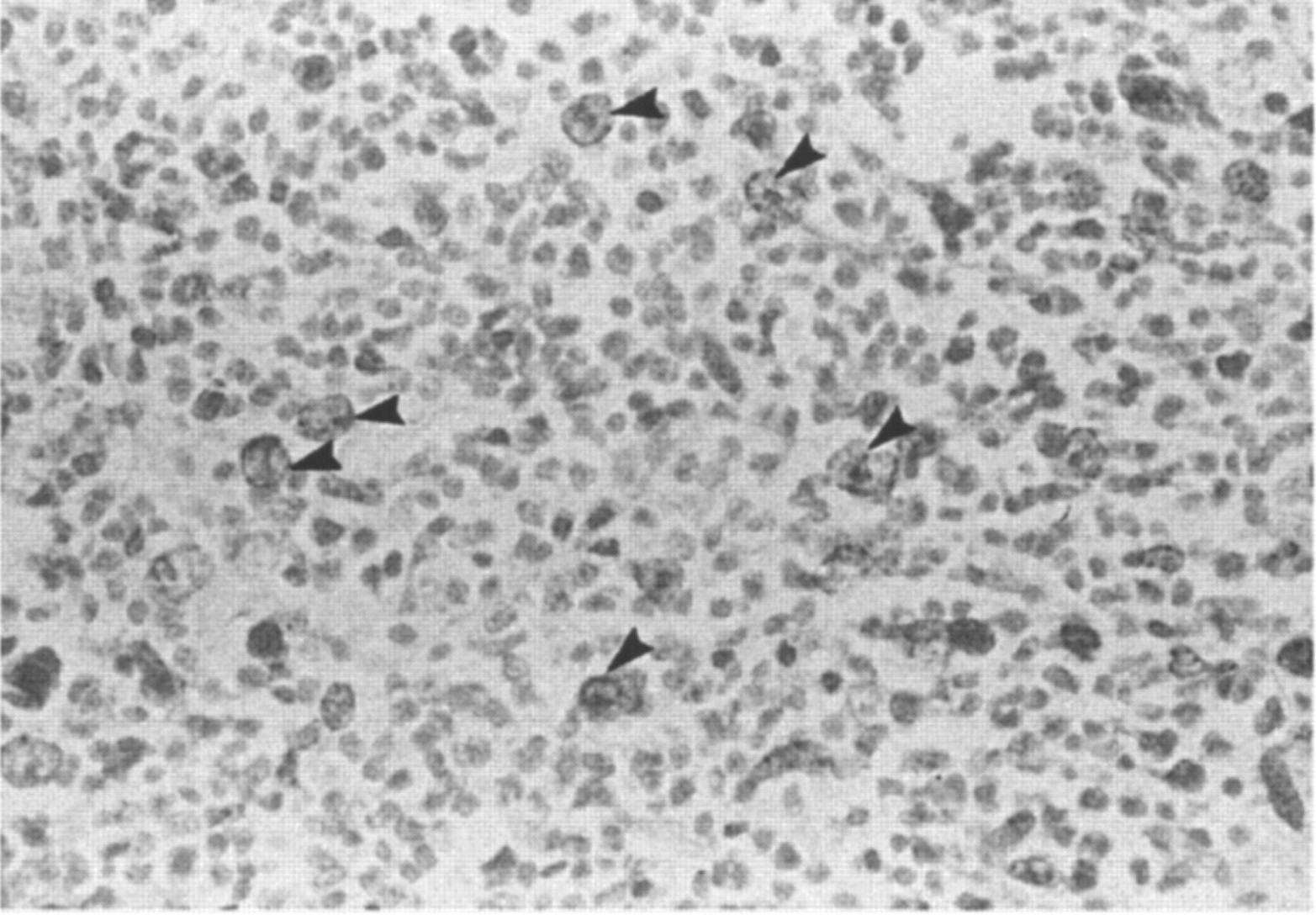
Note the large number of Bromodeoxyuridine stained cells (cycling cells) in the section (400×).
It is now believed that vaccines are not the only cause of sarcomas seen at injection sites. Virtually anything that produces local inflammation at the injection site may potentially be responsible for injection site sarcomas in susceptible cats, but vaccines are the only things that are given to most of the cat population with any frequency to make a good correlation. Reports of sarcomas developing at sites of antibiotic administration, etc. have been occasionally reported (personal communication).
Mechanisms other than inflammation have also been investigated. The potential role of viruses in the pathogenesis of vaccine site sarcomas has been partially studied. Ellis et al (1996) studied whether the feline leukaemia virus or the feline sarcoma virus were expressed in vaccine-site associated sarcomas in order to determine if there might be a viral mechanism giving rise to these sarcomas. One hundred and thirty vaccine site sarcomas were evaluated using polymerase chain reaction (PCR) and immunohistochemical staining but Ellis was unable to detect evidence of FeLV by either method. The possible role of the tumour suppressor gene P53 in feline sarcomas has been studied in a small number of sarcomas, and mutations in P53 were detected in only two of 10 fibrosarcomas studied (Mayr et al 1995).
Despite these possible other mechanisms, inflammation remains the most accepted hypothesis in the pathogenesis of vaccine-site associated sarcomas. For close to 100 years, investigators have observed that irritation, inflammation, and/or wounding are promoters of tumour development (Argyris 1985, Dvorak 1986, Sieweke & Bissell 1994). Clinical examples that support these findings are numerous (Dubielzig 1984, Dubielzig et al 1990, Woog et al 1983, Dunham 1972, Lieber et al 1986, Pound & McGuire 1978, Sanderson & Mackie 1979). Molecular studies have suggested that cancers develop as a result of a multitude of genetic changes and that the environment in which these changes occur influence the outcome of these genetic alterations, either promoting or suppressing transformed cells. Recent work indicates that oncogenes often need the interaction with other oncogenes or other factors in order to transform cells to a cancerous phenotype (Stocker et al 1990, Tato et al 1983). Four animal models provide additional insight as to possible mechanisms associated with the vaccine-induced tumours (v-src chicken model, bovine papilloma model, T cell lymphotropic retrovirus model, and v-jun transgenic mouse model). In these four animal models of virus- or oncogene-induced tumours, inflammation appeared to play a role in their pathogenesis. V-src infected chickens developed tumours at wound sites 10 to 15 days after injury (Dolberg et al 1985). Bovine papilloma virus elicits skin tumours in transgenic mice 8–9 months after birth in areas prone to scratching (Lacy et al 1986). The tat gene of human T-cell lymphotropic retrovirus type I induces mesenchymal tumours in transgenic mice at 3 months of age in areas prone to scratching (Nerenberg et al 1987). The v-jun transgenic mice developed tumours at 2–3 months of age only in areas where wounds have been made Schuh et al 1990). A clinical example of this phenomenon in the cat is the development of fibrosarcomas in areas of wounding in cats infected with the feline sarcoma virus (Hardy 1981). Of special interest in the possible prevention of vaccine-associated sarcomas is the recent work done in chickens infected with the Rous sarcoma virus indicating that if the post-wounding inflammation can be suppressed, tumours do not develop (Matins-Green et al 1994). If inflammation is considered a necessary antecedent to multiple events that ultimately lead to sarcoma development in some cats vaccinated with FeLV or rabies vaccines, then it would seem logical that measures that attempt to reduce post-vaccinal inflammation should be instituted. Although epidemiologic studies have thus far failed to identify specific brands of FeLV or rabies vaccines that may induce increased risk for the development of sarcomas, this may be due to the relatively low prevalence of sarcomas found in cats, large numbers of brands of FeLV and rabies vaccines administered to cats, or other factors associated with retrospective studies. Until a prospective study links specific brands of feline vaccines to tumour development, it may be useful to evaluate currently used rabies and FeLV vaccines for their capability of inducing local inflammation at vaccine sites.
The vaccine component most commonly thought to be associated with local post-vaccinal inflammation is the adjuvant (Vanselow 1987). Adjuvants are used in many but not all inactivated feline vaccines. Aluminium in the form of aluminium hydroxide or aluminium phosphate is a common component of vaccine adjuvants and is used in some FeLV vaccines and rabies vaccines (Vanselow 1987). Because aluminium has been identified to be present in post-vaccinal granulomas and in some sarcomas that developed at vaccine sites, it was considered a possible cause of tumour development (Hendrick et al 1992). However, aluminium may be only a marker of previous vaccination, and other vaccine components may induce inflammation or enhance the inflammatory process that results in sarcoma development in some cats. The specific role of aluminium, other adjuvants, adjuvant components, or vaccine antigens in inducing sarcomas in cats remains unknown at this time. Unlike human vaccines, carcinogenic assays are not required of United States veterinary vaccines.
In an attempt to identify vaccines most likely to induce local post-vaccinal inflammation, six inactivated feline vaccines (three rabies vaccines and three FeLV vaccines) were administered to 36 cats (six groups of cats, six per group) and were evaluated for evidence of local postvaccinal vaccine-site inflammation 21 days after subcutaneous administration (Macy DW, unpublished data). This study found that among cats vaccinated with inactivated rabies vaccines, local inflammatory reactions occurred in 80 to 100% of the vaccinates, depending on the brand of vaccine administered. The size of the local inflammation after vaccination was similar for all rabies vaccines, independent of whether the individual vaccine's adjuvant contained aluminium. The local post-vaccinal inflammatory lesions observed after rabies vaccination were approximately twice the size of the post-vaccinal inflammatory lesions found at vaccine sites in cats receiving any of the three feline leukaemia virus vaccines. Of the three feline leukaemia virus vaccines tested, the adjuvanted FeLV vaccines containing aluminium most consistently produced local post-vaccinal vaccine-site inflammation, followed by non-aluminium adjuvanted feline virus vaccines that produced measurable post-vaccinal reactions in only one of six vaccinates. Importantly, the FeLV vaccine that contained no adjuvant produced no measurable local inflammation at vaccine sites. A quantitative morphologic evaluation of the cellular component contributing to this inflammatory response is currently underway.
In an attempt to develop a cost-friendly model, we repeated the above experiment in rats and obtained identical results. This model has allowed us to look at other feline vaccines and routes of vaccine delivery. We have recently evaluated a non-adjuvanted 1-year rabies vaccine and a non-adjuvanted 3-year rabies vaccine and have found they produce either little or no inflammation 21 days post administration.
Some investigators have suggested that IM-administered vaccines do not produce tumours, and that massaging vaccine sites after vaccine administration may prevent subsequent tumour development. We evaluated IM and SQ routes of administration and found no significant reduction in inflammation by administering a vaccine intramuscularly. Further, we investigated the effect of massaging vaccine sites, and found that massaging a vaccine site only changes the shape but not the volume of the post-vaccinal inflammatory responses.
Although local inflammation may be induced in nearly 100% of vaccinates with certain rabies and FeLV vaccines, tumours only develop in approximately 1/1000 of these vaccinates (Macy DW, unpublished data). This fact, along with the observations that some cats develop sarcomas at every vaccine site, some treatment failure lesions distant to the primary tumour site contain adjuvant, and that many related cats have been found to be affected with vaccine-associated sarcomas, suggests an individual inherited susceptibility or genetic defect may play a role in the pathogenesis of these sarcomas (Macy DW, unpublished data). An investigation of the cytogenetics of cats that develop granulomas versus those that have vaccine-site associated sarcomas is underway at our institution to study this possibility.
Recommendations for preventing or reducing the incidence of vaccine-associated tumours in cats are controversial. Recommendations include a change in vaccination site location, decreased use of polyvalent vaccines, use of non-adjuvanted vaccines, avoiding the use of aluminium based adjuvants, and not over-vaccinating, among others (Macy & Hendrick 1996, [AVMA/VAFSTF web site: http://www.avma.org/vafstf/default.htm] Advisory panel on feline vaccines 1998).
The most important recommendation for prevention of vaccine-associated sarcomas would appear to be not to overvaccinate. Kass et al (1993) has clearly shown that the risk of vaccine-associated sarcomas increases with the number of vaccines administered. It is also clear from recently published articles by Scott et al (1997) that the duration of immunity of many of our feline products is longer than 1 year and an every 3-year program should be instituted for many vaccines for infectious agents of cats. Although the triennial vaccination program has been endorsed by the American Association of Feline Practitioners (Macy & Hendrick 1996, [AVMA/VAFSTF web site: http://www.avma.org/vafstf/default.htm] Advisory panel on feline vaccines 1998), it has not been warmly received by general practitioners in the United States. With respect to rabies vaccination, annual vaccination for rabies with a 3-year rabies product should be discouraged (Macy & Hendrick 1996, [AVMA/VAFSTF web site: http://www.avma.org/vafstf/default.htm] Advisory panel on feline vaccines 1998). Most 1-year rabies vaccines used in the United States are really a 3-year rabies vaccines. Unfortunately, in the United States, a 3-year rabies vaccine may be relabeled and sold as a 1-year product. The indiscriminate use of FeLV vaccine should be stopped because epidemiologic data suggest it has the greatest association with the development of injection site sarcomas in cats. The recent article by Romatowski (1997) casts serious doubt whether vaccinating cats in single cat households has had any impact on the incidence of FeLV in that population. If FeLV vaccines are to be used at all, they should be reserved for high risk cats and given to the young prior to 12 weeks of age when the chance of developing persistent FeLV infection if exposed is 85% (Hoover et al 1976). Experimental studies have indicated that once cats are over 6 months of age, only 15% of exposed cats will develop persistent FeLV infections (Hoover et al 1976). Given the potential risk of sarcoma development (1–10/10 000), a veterinarian should really question the benefit of FeLV vaccination in individual cats. A veterinarian may be doing more harm than good in vaccinating against FeLV annually in the single cat household population. Fortunately, very few veterinarians in the United States now recommend FeLV vaccination for strictly indoor cats. The frequency of revaccination for FeLV should be based on duration of immunity studies, which are unfortunately still almost all US veterinary vaccines.
Vaccine site location recommendations have also recently been changed. The National Feline Vaccine-Associated Sarcoma Task Force recommends that no vaccine be given in the interscapular space, that rabies vaccine be administered subcutaneously in the distal right rear leg, that FeLV vaccine be administered subcutaneously in the distal left rear leg, and that all other vaccines be administered subcutaneously in the right shoulder (Macy & Hendrick 1996, [AVMA/VAFSTF web site: http://www.avma.org/vafstf/default.htm] Advisory panel on feline vaccines 1998). It appears from our research and reports by others that intramuscular and subcutaneous administration both result in local inflammation and tumour production. Subcutaneous sites are recommended for all vaccines since they will result in earlier detection of these growths if they occur.
The reasoning behind these vaccine site administration recommendations is not based so much on prevention but rather on earlier diagnosis and potentially a higher cure rate when treated surgically.
The issue of monovalent and polyvalent vaccines is still controversial. Kass (1993) found a significant positive trend in risk with an increasing number of vaccines. However, Hendrick et al found no association between vaccination-site and nonvaccination-site tumours in the number of vaccinations given simultaneously. Although ambiguity exists, it seems prudent to now recommend that vaccines be administered at different sites and not to use polyvalent vaccines. The current practice that all cats should receive all available feline vaccines annually is being seriously challenged by many veterinarians in the United States today, but still remains the way most practitioners vaccinate cats.
The use of vaccines containing aluminium-based adjuvants is controversial. The recommendation not to use multiple-dose vials for aluminium-adjuvanted vaccines appears less controversial. The fact that aluminium is in suspension and not solution makes it is difficult if not impossible to obtain an equal concentration of vaccine adjuvant and attached vaccine protein every time a dose is drawn up into a syringe (Macy DW, unpublished data). Recent work at our institution using atomic absorption spectrophotometry confirmed this hypothesis, thus only single-dose vaccine vials should be used if giving aluminium-adjuvanted vaccines (Macy DW, unpublished data). Because aluminium-adjuvanted products are the most consistent producers of vaccine-site inflammation, in my opinion, they should be avoided if possible! However, non-aluminium-based adjuvants have also been linked to vaccine-site tumours, and may be equally unsafe. More research is clearly needed to determine the real risk of aluminium adjuvanted-vaccine products in the induction of vaccine-site associated sarcomas.
It would appear that the problem of vaccine-site associated sarcomas will be with us for some time, and the question of what to do with post-vaccination lumps is a practical one. Some rabies and FeLV vaccines produce post-vaccinal lumps in 100% of the vaccinates but, fortunately, it has been observed that most resolve in 2–3 months. Very few vaccine-associated tumours occur sooner than 3 months following vaccination. Given these observations, we currently recommend that any vaccine site lumps present after 3 months from the time of vaccination be removed, but only after a biopsy. A biopsy will determine the magnitude of the surgery, ie, lumpectomy versus radical surgery. We do not recommend excising the mass prior to biopsy. Attempts at simple excision of these tumours is seldom curative and ultimately leads to local recurrence with a more difficult second surgical attempt (Macy & Bergman 1995). Even attempts at aggressive wide surgical excision are often incomplete and result in a 30–70% failure rate (Hershey et al 1997, Davidson et al 1997). There appears to be an advantage to refer these cases to a qualified surgeon before the second surgery is contemplated. After a second surgical intervention has been attempted, there appears to be no improvement in survival with referral to specialist surgeons. Rear leg amputation has a higher rate of cure than surgery in the interscapular space for vaccine-site associated sarcomas. Given the incomplete removal of these tumours even with aggressive surgery, radiation is often used before or after surgery. One study's interim survival analysis indicates a doubling of survival when radiation is included ([Macy DW, unpublished data], Hershey et al 1997). Although the combination of surgery and radiation has increased tumour control rates in some studies, a significant number of cats still fail to respond to this combination. Several chemotherapy agents, including carboplatin, doxorubicin, cyclophosphamide, Cytoxan and vincristine, have been used in cats with vaccine-associated tumours (Ogilvie & Moore 1995). Most chemotherapeutic attempts result in partial responses, but some complete responses have been observed with these drugs. Immunomodulators have been used systemically or intralesionally, but response has been limited (Kent 1993). Although the vast majority of vaccine-site associated sarcomas are only locally invasive, approximately 10–20% will metastasize to the lungs or other sites (Briscoe et al 1995, Esplin & Campbell 1995, Rudmann et al 1996).
The cost of the treatment for vaccine-site associated sarcomas can be significant and run into many thousands of dollars. The ethics of the veterinary profession and how we manage this problem are being watched by many, including the media. How we act on this issue is likely to have a profound effect on how our profession is perceived by the public for some time in the future.
Given the need for vaccination against rabies to prevent a lethal disease in cats and humans, there would appear to be a need to compensate owners for expenses associated with the treatment of sarcomas that develop after the administration of mandated vaccines like rabies. Perhaps it is time that the veterinary profession consider a vaccine injury act similar to the 1986 Childhood Vaccine Injury Act in use for humans injured by mandated vaccines such as diptheriapertussis-tetanus (DPT) and measles-mumps-rubella (MMR) in the United States (Macy 1998). More legislative support is also needed and required in truth in labeling of United States' veterinary vaccines that have been associated with vaccine-site sarcoma development. Currently, the fact that a 3-year rabies vaccine may be relabeled as a one-year product seems unacceptable. Equally unacceptable is the fact that FeLV vaccines are recommended by manufacturers to be given annually without any evidence of duration of immunity.
Unlike European vaccines, no minimum duration of immunity must be established prior to licensure for veterinary vaccines, so regardless if the duration of immunity is three months or a lifetime, US vaccine manufacturer label recommendations are ‘annual revaccination’. A critical look at duration of immunity using our understanding of immunity derived from previous natural infection, the prevalence of disease in cats previously vaccinated for the disease, as well as serologic data when appropriate, should help the profession establish more realistic booster intervals and a reduction in the number of vaccines administered and, hopefully, a reduction in vaccine-associated sarcoma development.
