Abstract
Abstract
Purpose
Kingella kingae is increasingly recognized as a pathogen of osteoarticular infections (OAI) below the age of 2 years. It was reported that bones and joints which are rarely infected by other pathogens were frequently invaded by K. kingae. Based on a series of six cases, we present the typical clinical and paraclinical manifestation of K. kingae infections of the sternum and sterno-manubrial joint.
Methods
A review of the clinical, laboratory, radiological, microbiological, and molecular data of six consecutive children admitted to a paediatric unit for OAI of the sternum was done.
Results
Culture alone allowed for the detection of K. kingae as the responsible pathogen in three cases, molecular methods in the three other cases. Clinical and laboratory findings, as well as imaging methods, proved to be useful in the diagnostic process.
Conclusion
Our findings suggest that infections of the lower sternum and the junction between the manubrium and the xyphoid process are typical, if not pathognomonic, for the organism. A respective diagnostic and therapeutic protocol was established.
Keywords
Introduction
Kingella kingae has been increasingly recognized as a cause of paediatric osteoarticular infections (OAI) since the early 1980s [1–4]. Meanwhile, numerous skeletal manifestations of invasive K. kingae infection have been reported in the literature, owing to increased awareness, improved detection of the organism, or to actual rise in prevalence [4–7].
Herein we report the clinical, radiological, microbiologic, and molecular findings from a series of six children admitted to our institution between January 2004 and August 2006 for localized OAI of the lower sternum caused by this emerging paediatric pathogen.
Case reports
The distribution of patients according to their symptoms and paraclinical findings is shown in Table 1. Of the six patients, two were boys, four were girls, and the male-to-female ratio was 1:2. Patient median age was 11.8 months (range 10–15 months). Among the six patients, none had an underlying disease. The average interval from symptom onset to hospitalization was 10.5 days (range 3–15 days). All six infections in our series occurred in autumn (one case) or winter (five cases).
Patient characteristics
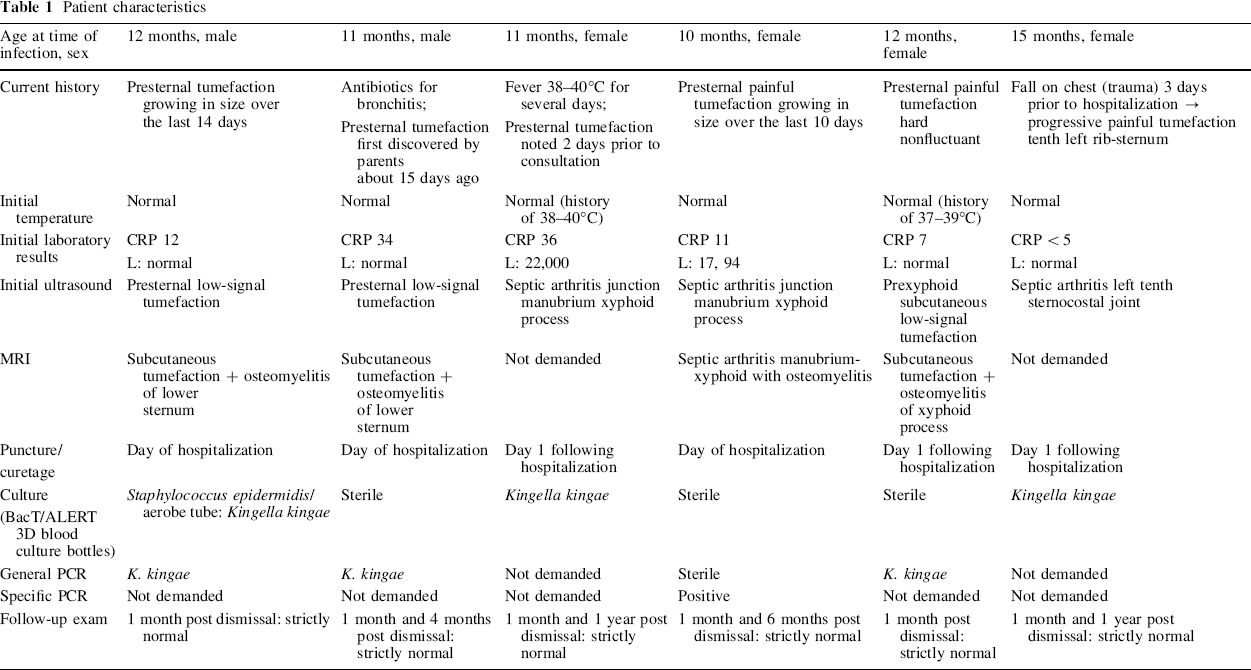
Clinical data
In all six patients, the chief complaint on admission was a painful, hard, nonfluctuating presternal mass that had gained in size over the past few days (Fig. 1). One patient had a history of a blunt trauma due to a fall on the chest from a standing height 3 days prior to admission. Only one patient, who had an upper respiratory tract infection treated by amoxicillin and clavulan acid, had received antibiotics before admission. Body temperature was normal on admission in all cases, but two patients had history of relapsing-remittent fever peaks up to 39°C over the past few days prior to hospital admission.
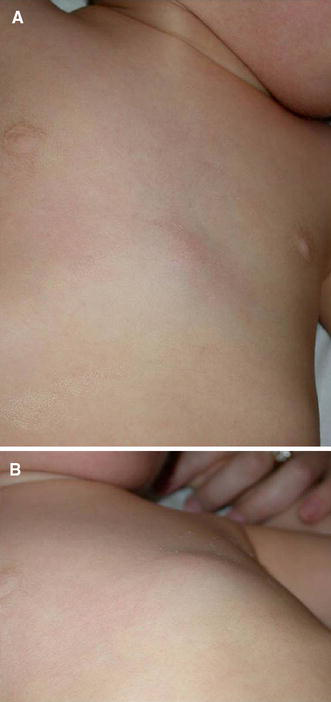
Ten-month-old girl presenting a painful, hard, nonfluctuating presternal mass that had gained in size over the past few days
Laboratory data
At presentation, average white blood cell (WBC) count was 14.67 g/L (range 8.01–22.80 g/L) and was elevated only in two patients. C-reactive protein (CRP) was elevated in five of six patients and averaged 16.7 mg/L (range 5–34 mg/L). Platelet count averaged 478,000 G/L (range 349,000–663,000 G/L).
Radiological data
Plain radiographs of the sternum were normal in all cases. Ultrasound scan revealed a circumscribed noncapsulated low-signal tumefaction of approximately 20 × 10 × 5 mm in size, located subcutaneously to the lower manubrium in two, subcutaneously to the xyphoid process in one, at the junction of the manubrium and the xyphoid process in two, or at the junction of the tenth left rib and the manubrium in the patient with a history of blunt chest trauma, respectively. In all cases in which magnetic resonance imaging (MRI) was performed, ultrasound findings were confirmed. In addition, lytic lesions of the lower manubrium were detected by MRI imaging in two cases and of the xyphoid process in two cases, showing a circumscribed area with signal augmentation in the T2 sequence and increased gadolinium uptake in the T1 sequence (Fig. 2).
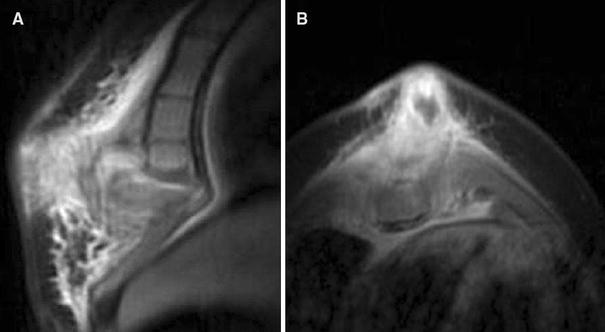
MRI imaging of the same patient, showing subcutaneous tumefaction, arthritis at the junction of the manubrium and xyphoid process with lytic lesions of the lower manubrium and xyphoid process
Microbiologic and molecular methods
Diagnostic aspiration was performed on the day of admission in three cases, and on the day following admission in three. Samples were directly inoculated by the surgeons into aerobic and anaerobic BacT/ALERT 3D blood culture bottles (bioMérieux). Three of them yielded positive culture for K. kingae in aerobic blood culture bottles. Universal 16S rDNA polymerase chain reaction (PCR) as described elsewhere [7] was obtained in all three cases in which culture was negative. Significant sequence homology (99–100%) with the rDNA sequence of K. kingae was found in two cases. K. kingae-specific real-time PCR as recently described [1] was obtained for one case in which universal 16S rDNA PCR was negative and revealed significant sequence homology (99–100%) with the rDNA sequence of K. kingae (100%).
Treatment
After diagnostic aspiration and surgical excochleation, all patients received intravenous treatment with a second-generation cephalosporin combined with an aminoglycoside for 48 h, followed by intravenous second-generation cephalosporin monotherapy for 8 days and the same antibiotic orally after dismissal for another 3 weeks.
Follow-up and outcome
Follow-up 3 and 6 weeks after dismissal included clinical and laboratory controls as well as ultrasound scan. All six patients were completely healthy and did not present any sequelae of their disease. Local healing was uneventful in all cases. Laboratory results were strictly normal in all cases, and ultrasound scan showed frank diminution of the described lesion. One-year follow-up exams took place in three of the six patients who continued follow-up visits in the outpatient clinic, and were strictly normal in all cases.
Discussion
Kingella kingae has been identified as a pathogen in cases of meningitis, endocarditis, and spondylodiscitis, and is increasingly recognized as a cause of osteoarticular infections (OAI) in young children [4, 8–10]. Moreover, in a recent study using K. kingae-specific real-time PCR, K. kingae was placed as the leading cause of OAI in young children [1]. It has also been reported that K. kingae tends to cause infections in unusual bones and joints, such as the clavicle, calcaneus, talus, sternoclavicular joint, and wrist [11–15]. During the study period, routine use of both blood cultures and molecular methods allowed for identification of K. kingae as the responsible pathogen in 39 children admitted to our institution for OAI. Anatomical distribution included 27 cases of arthritis, 7 cases of osteomyelitis, and 5 cases of spondylodiscitis. Interestingly, the most common site of osteomyelitis was the sternum. OAI of the sternum have been mentioned in previous reports but was scarcely specifically elaborated upon [4]. To the best of our knowledge, a single case report of a presternal soft tissue infection caused by K. kingae in a 3-year-old child can be found in the literature [16].
Kingella kingae is a common component of the normal oropharyngeal flora in early childhood. The seasonal predominance of invasive infections with this pathogen in autumn and winter, as reported in this and other studies, may thus be attributable to concomitant viral infections [4].
The age distribution of our six patients concurs with that of previous reports, K. kingae OAI being found in a defined age group (6–36 months) [4, 7]. In the presented patient group, average age was 11.8 months (range 10–15 months).
Kingella kingae infection can be difficult to diagnose as clinical signs are usually mild. In the presented patient group, body temperature was noted to be normal at time of presentation in all cases (while two had a history of relapsing-remittent fever peaks over the past few days prior to hospital admission), and local symptoms were mild. CRP concentrations and WBCs may be moderately elevated.
Imaging methods proved to be useful. While plain radiographs showed no significant alterations, ultrasound scans allowed for detection of soft tissue infections in three patients and osteoarthritis in three patients, respectively. Performed in four cases, MRI imaging studies revealed localized osteomyelitis of the lower manubrium in two of these patients and of the xyphoid process in two others. All these data were confirmed intraoperatively and are described in Table 1.
The importance of appropriate culture media that can detect K. kingae, namely aerobic blood culture bottles, has been pointed out [17]. Moreover, systematic use of molecular methods on culture-negative samples has been demonstrated to increase the detection of K. kingae in OAI [1, 7]. In this study, aerobic blood culture bottles allowed for detection of K. kingae as the responsible pathogen in three cases, and molecular methods (universal 16S rDNA PCR [7], as well as K. kingae-specific real-time PCR [1]) for the detection of K. kingae in the three other cases. Our awareness toward this type of infection in this narrow age group has led to the establishment of the described progressive diagnostic protocol, which we believe to be crucial for detection of K. kingae.
Treatment of our patients was based on diagnostic aspiration, surgical excochleation, and intravenous antibiotic therapy over a period of 10 days, followed by the same antibiotic orally for 3 weeks as described above. Outcome was favorable in all cases after 1 month of follow-up. Upon identification of the characteristic lesion (Fig. 1) in a typical clinical context and confirmation by imaging methods, empiric application of a proven antibiotic treatment protocol without surgical intervention will be considered in the future.
The reported series of K. kingae OAI of the sternum represents a homogenous clinical entity and suggests that infections of the lower sternum and the junction between the manubrium and the xyphoid process are typical, if not pathognomonic, for the organism.
