Abstract
Abstract
Purpose
The 22q11.2 deletion syndrome is a common genetic syndrome with a wide spectrum of abnormalities. We have previously described multiple anomalies of the upper cervical spine in this disorder. The objective of this study was to use advanced imaging to further define the morphology of the cervical spine and spinal cord in the 22q11.2 deletion syndrome, with a comparison to age-matched controls.
Methods
A total of 32 patients with a 22q11.2 deletion underwent advanced imaging (computed tomography/magnetic resonance imaging; CT/MRI) of the cervical spine. In 27 patients, space available for the cord (SAC); the sagittal diameter of the vertebral body, spinal canal, cerebrospinal fluid (CSF), and spinal cord; and the cross sectional area of the spinal canal, CSF, and spinal cord were measured at each cervical level and compared to 29 age-matched controls. Statistical analysis was performed and potential implications were hypothesized.
Results
In 22q11.2 patients, advanced imaging identified 40 pathologies not evident on plain radiographs with potential mechanical and/or neurological implications. These patients also had significantly smaller values (P ≤ 0.05) of the following parameters at one or more cervical levels, relative to age-matched controls: width of the vertebral body, spinal canal, CSF, and spinal cord; area of the spinal canal, CSF, and spinal cord. Neurologic symptoms were observed in 4/32 patients, with one patient requiring surgical intervention.
Conclusions
Advanced imaging of the cervical spine can detect findings not evident on plain radiographs in the 22q11.2 deletion syndrome. CT and/or MRI may be indicated when there is a high index of suspicion for clinical instability or neurologic compromise in order to rule out dynamic encroachment or impending neurologic sequelae. Spinal canal and spinal cord dimensions are reduced in these patients relative to controls with currently unknown clinical significance.
Introduction
The deletion of chromosome 22q11.2 is the most commonly known interstitial chromosome deletion found in humans, with an estimated prevalence of 1 in 4,000–6,000 live births, although the true prevalence is likely to be higher [1–4]. The 22q11.2 deletion syndrome is a disorder that encompasses a wide spectrum of abnormalities, including orthopedic anomalies [1, 4–14].
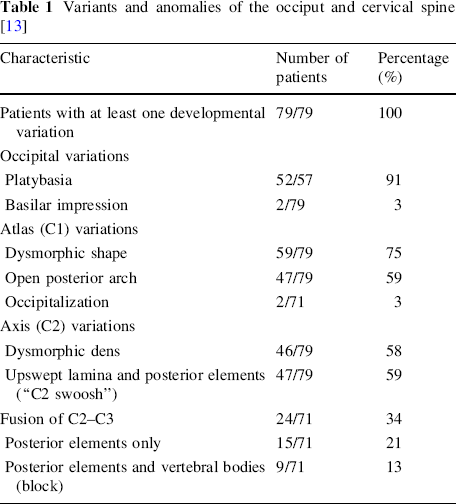
We recently described a total of 279 variants or anomalies of the occiput and cervical spine using plain radiographs in 79 consecutive patients with this chromosome deletion: 54 of the occiput (52 platybasia, two basilar impression), 108 of C1 (59 dysmorphic shape, 47 open posterior arch, two occipitalization), 93 of C2 (46 dysmorphic dens, 47 upswept lamina and posterior elements), and 24 fusions of C2–C3 (Table 1) [13]. Increased segmental motion at multiple cervical levels was also noted [13] and at least one abnormality of the cervical spine was observed in every patient (mean of 2.9 per patient).
Variants and anomalies of the occiput and cervical spine [13]
Central nervous system (CNS) anomalies of the cervical spine, such as Chiari type I malformations, have previously been reported in the 22q11.2 deletion syndrome using advanced imaging [5, 7, 9, 11, 15]. In addition, we have recently shown decreased spinal canal and spinal cord dimensions in other syndromic patients with cervical spine abnormalities [16]. The 22q11.2 patients may show similar findings. Advanced imaging such as computed tomography (CT) and magnetic resonance imaging (MRI) may be needed to adequately define the abnormalities of the cervical spine in the 22q11.2 deletion syndrome and their clinical effects. With multiple abnormalities already known in this patient population, the syndrome also represents an ideal cohort to examine in order to better understand cervical spine development. Therefore, the purpose of this study was to use advanced imaging (CT and/or MRI) to further define the morphology of the cervical spine and spinal cord in the 22q11.2 deletion syndrome and to postulate the possible clinical implications.
Materials and methods
Between 31st October 2001 and 30th June 2005, a cohort of 87 consecutive patients referred from the “22q and You Center” in the department of Clinical Genetics at the authors’ institution underwent clinical and radiographic evaluation of the cervical spine. Institutional review board (IRB) approval and informed consent were obtained. The clinical evaluation included a detailed history and physical examination focusing on neurologic complaints. Initial radiographic evaluation included lateral plain radiographs in neutral, flexion and extension, anteroposterior (AP) radiographs, and open-mouth odontoid views. All patients less than one year of age were excluded from the study because cervical spine findings in this age group could not be accurately assessed on plain radiographs.
Based on the presence of any neurologic signs or symptoms or radiographic evidence of possible cervical spine instability (increased segmental motion on flexion/extension views), patients underwent advanced imaging with CT and/or MRI of the occiput and cervical spine. A total of 24 of the original 79 patients from our plain radiographic study [13] and eight additional patients met the criteria for inclusion and underwent advanced imaging. Multiplane imaging, including axial CT with coronal and sagittal reconstructions and axial, coronal, and sagittal MRI, was used to confirm plain radiographic findings and detect additional abnormalities. Dynamic CT and MRI studies were not performed. Advanced imaging studies were evaluated by all four authors independently, followed by a group review of the findings. In cases where there was a disparity in the findings, agreement was reached by consensus.
Abnormalities initially noted on plain radiographs were defined as we have described previously [13, 17–29] and included platybasia, basilar impression, dysmorphic C1, open posterior arch of C1, occipitalization of C1, dysmorphic dens, upswept lamina and posterior elements of C2 (termed the “C2 swoosh” [13]), partial or complete fusion of C2–C3, and increased segmental motion at one or more levels of the upper cervical spine, including occipitoatlantal, atlantoaxial, C2–C3, and C3–C4. Segmental motion or instability was not assessed via advanced imaging in the current study because dynamic CT and MRI studies were not performed.
Digital advanced imaging was available for 27 of the 32 patients to evaluate the spinal canal and spinal cord dimensions in the cervical spine. Five patients had advanced imaging performed at outside facilities and digital imaging was not available for making precise measurements of these dimensions. These measurements included the space available for the cord (SAC); the sagittal diameter of the vertebral body, spinal canal, cerebrospinal fluid (CSF) with contained neural elements, and spinal cord; and the cross sectional area of the spinal canal, CSF with contained neural elements, and spinal cord (note: “CSF with contained neural elements” will be referred to only by “CSF” in the remainder of the manuscript). These diameter (mm) and cross sectional area (cm2) measurements were made on CT and MRI as described previously by Prasad et al. [30]. On CT, the SAC was measured on axial images at the mid-vertebral level of C1 from the posterior aspect of the odontoid to the anterior aspect of the posterior arch of the atlas [19, 31]. The cross sectional area of the spinal canal was measured on axial images at the mid-vertebral level from C2 to C7 (C1 was not measured because of inaccuracies imposed by the presence of the odontoid). On MRI, the T2-weighted sagittal image showing the midpoint of the spinal cord in each patient was used to measure the diameter of the vertebral body, spinal canal, CSF, and spinal cord at the mid-vertebral level from C1 to C7 (odontoid width measured as vertebral body diameter at C1). T2-weighted axial magnetic resonance images were used to measure the cross sectional area of the CSF and the spinal cord at the mid-vertebral level from C1 to C7. The cross sectional area of the CSF was measured instead of the bony canal because the limits of the bony canal are not easily visualized on MRI, while the CSF column is clearly distinguishable on T2-weighted images [30]. The SAC was measured on T2-weighted axial magnetic resonance images in the same manner as CT [19, 31]. All measurements were performed using the measurement feature of the digital imaging program on which the studies were stored at our institution (IDX Imagecast, GE Healthcare). This program instantaneously calculates both diameters (mm) and cross sectional areas (cm2) (Fig. 1). Measurements were compared to values made on the same imaging study (CT or MRI) of 29 age-matched controls without cervical spine pathology identified from our institutional trauma registry.
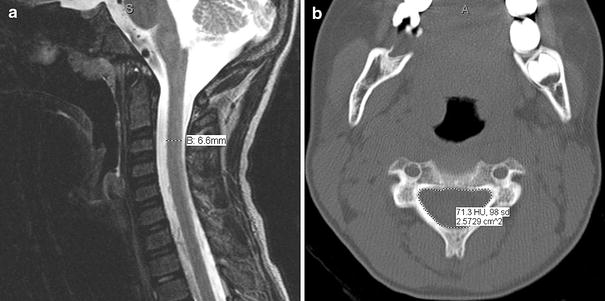
Sagittal T2-weighted magnetic resonance image (
Several additional findings noted on advanced imaging but not initially seen on plain radiographs were defined. An open anterior arch of C1 and open anterior or posterior arches of cervical levels below C1 were detectable on the axial views of either CT or MRI. Lateral mass subluxation at C1 was detectable on the coronal views of either CT or MRI as widening of the space between the lateral masses and the odontoid. Cervical spinal canal encroachment was defined on either CT or MRI as a reduction in the SAC of ≤13 mm [31, 32]. Encroachment may be due to static and/or dynamic factors. Static encroachment may occur from bony narrowing of the spinal canal (stenosis), the presence of a space-occupying mass in the canal (i.e., Chiari malformation), or protrusion of the odontoid through the foramen magnum (basilar impression). Dynamic encroachment may occur as the result of increased segmental motion or cervical spine instability [31, 32]. Cervical spinal cord impingement was assessed on MRI and is defined by an identifiable indentation on T2-weighted sagittal views, either anteriorly or posteriorly, in the spinal cord in the region of the cervical spine.
Statistical methods
Statistical analyses were performed using univariate analysis of covariance (ANCOVA) to compare spinal canal and spinal cord dimensions between the 22q11.2 patients and the age-matched controls. Age was chosen as the covariate in the model in order to allow for more accurate comparisons, as spinal canal and spinal cord dimensions (both diameters and cross sectional areas) are expected to increase with age in this pediatric patient population [33]. The age-adjusted means of each measurement at each cervical level on CT or MRI were compared between the 22q11.2 patients and the age-matched controls with a statistically significant difference set at P ≤ 0.05.
Results
A total of 32 patients with a genetically documented 22q11.2 deletion met the criteria for advanced imaging of the cervical spine and underwent CT and/or MRI. The 16 male and 16 female patients had a mean age of 8.94 years. Nine patients underwent CT alone, 14 underwent MRI alone, and nine underwent CT and MRI. Plain radiographs were not available for the comparison of findings in one patient.
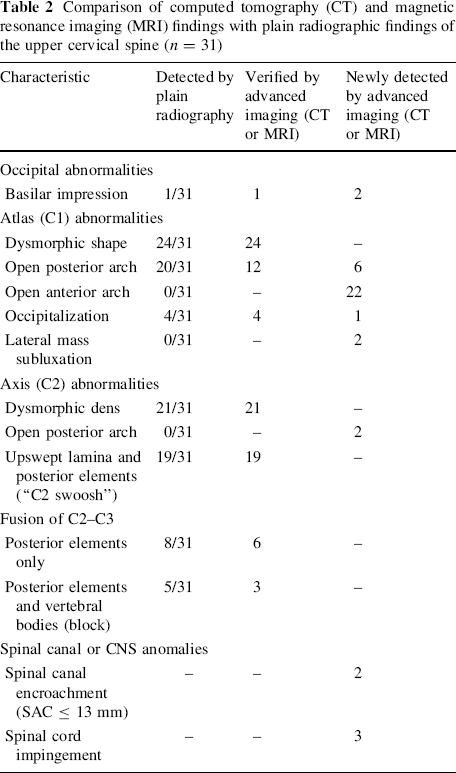
Advanced imaging identified 40 additional pathologies not evident on plain radiographs (Table 2; Figs. 2–4). No CNS abnormalities were noted (syrinx, Chiari type I malformation, tumor, etc.). There were a few instances where advanced imaging was unable to confirm plain radiographic findings. Three cases of an apparent open posterior arch of C1 on plain radiographs were found to be closed on CT. Five other patients with an apparent open posterior arch of C1 on plain radiographs who underwent MRI only also did not have this finding confirmed. Two cases of C2–C3 block fusion and two cases of C2–C3 posterior fusion on plain radiographs were also not confirmed by MRI alone.
Comparison of computed tomography (CT) and magnetic resonance imaging (MRI) findings with plain radiographic findings of the upper cervical spine (n = 31)
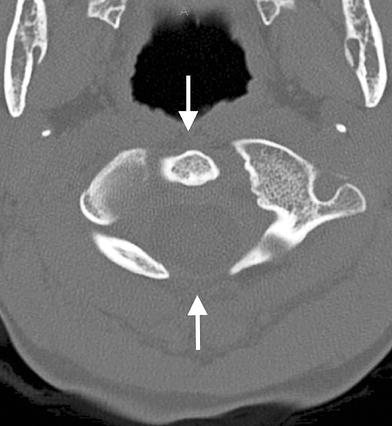
Axial CT image depicting open anterior and posterior arches of C1 (arrows)
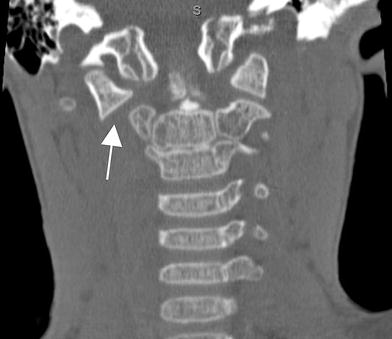
Coronal CT image depicting lateral mass subluxation of C1 on the right (arrow)
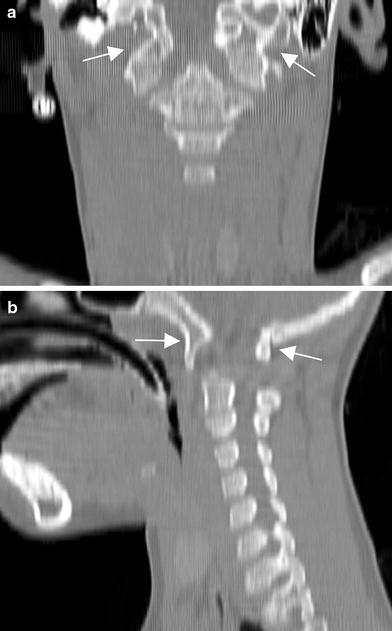
Coronal (
Digital advanced imaging was available for 27 of the 32 patients with a 22q11.2 deletion to evaluate spinal canal and spinal cord dimensions in the cervical spine. The 14 male and 13 female patients had a mean age of 7.78 years. Eight patients underwent CT alone, eleven underwent MRI alone, and eight underwent CT and MRI. The sagittal diameter and cross sectional area measurements were compared to findings in 29 age-matched controls (15 male and 14 female patients with a mean age of 8.19 years). Ten control patients had CT studies for comparisons and 19 control patients had MRI studies for comparisons.
Age-adjusted mean values of each diameter and cross sectional area measurement at each cervical level on CT or MRI were compared between the 22q11.2 patients and the age-matched controls (Table 3). All but five measurements (CT SAC, CT canal area at C2, MRI sagittal spinal cord width at C6 and C7, MRI axial CSF area at C1) showed smaller mean values in the 22q11.2 patients compared to age-matched controls. Using ANCOVA with age as the covariate, the following parameters showed statistically significant (P ≤ 0.05) differences in their means: CT canal area at C4; MRI sagittal body width at C1–C6, MRI sagittal canal width at C1–C4, MRI sagittal CSF width at C1–C7, MRI sagittal spinal cord width at C2–C3; MRI axial CSF area at C3–C4; and MRI axial spinal cord area at C4. In every comparison with a statistically significant difference, the control cohort had a higher mean value than the 22q11.2 patients.
Comparison of spinal canal and spinal cord measurements in the cervical spine between 22q11.2 patients and age-matched controls
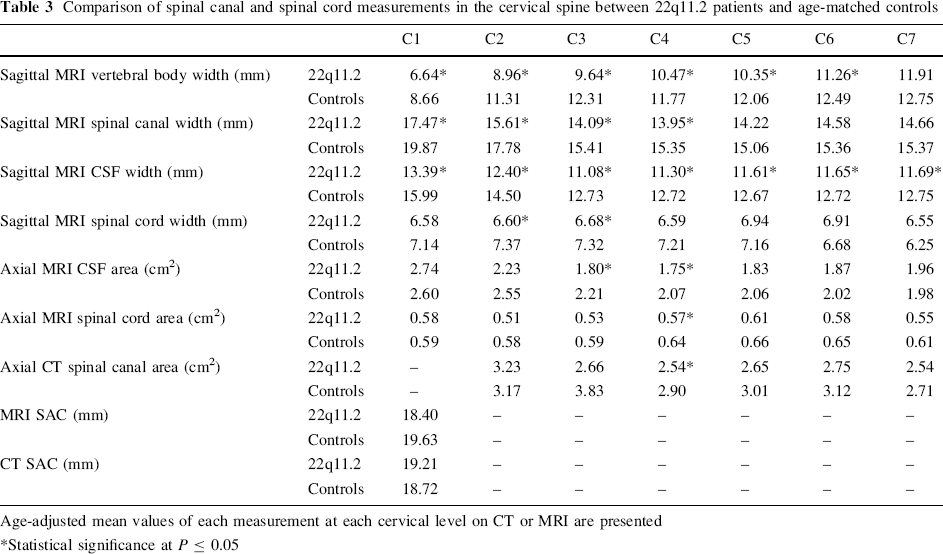
Age-adjusted mean values of each measurement at each cervical level on CT or MRI are presented
Statistical significance at P ≤ 0.05
As we have noted previously [13], despite the high frequency of cervical spine abnormalities in the 22q11.2 patients, the majority of patients are asymptomatic in childhood. In this study, four of the 32 (12.5%) patients showed evidence of some neurologic signs or symptoms, such as developmental delays, coordination and fine motor difficulties, subtle gait disturbances, or radiculopathy. One of these four patients presented with progressive upper motor neuron signs, weakness, and myelopathy. CT and MRI demonstrated canal encroachment at C1 with spinal cord impingement posteriorly at that level, and the patient underwent decompression with resolution of his symptoms (Fig. 5). The other three patients did not have evidence on advanced imaging of canal encroachment or spinal cord impingement in the cervical spine and have not had progression of their symptoms, but remain under close observation, with sports restrictions for diving, tumbling, and heavy contact sports.
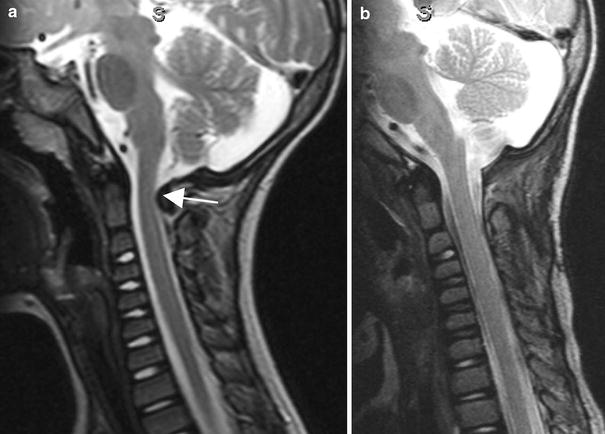
Sagittal T2-weighted MRI images of a 34-month-old male patient with symptomatic spinal canal encroachment and spinal cord impingement.
Discussion
The purpose of the current study was to use advanced imaging to further define the morphology of the cervical spine and spinal cord in the 22q11.2 deletion syndrome and to postulate the potential clinical significance of these findings. Spinal canal and spinal cord dimensions in the 22q11.2 patients were compared to an age-matched control group in order to determine differences from the norm. Our initial report of 79 patients was the first in the literature with an extensive radiographic description of abnormalities of the cervical spine in patients with the 22q11.2 deletion syndrome [13]. The CT and MRI findings in the current study confirmed most of the anomalies that we detected on plain radiographs. In addition, advanced imaging detected 40 additional pathologies not previously observed. An open anterior arch of the atlas was the most commonly detected new finding (22 patients), and was not previously found on plain radiographs in any of the patients. We also previously reported an open posterior arch of C1 to be a very common finding in these patients (59%) [13], but six new cases were detected in the current study that were missed on plain radiographs. Overall, 15 patients in the current study had both open anterior and posterior arches of C1 detected by advanced imaging. Two of these patients were noted to have evidence of C1 lateral mass subluxation on CT, which may be a consequence of both the front and back of the atlas being open. Other abnormalities newly detected with advanced imaging in this study included a case of occipitalization of C1 with basilar impression and one case of isolated basilar impression.
The clinical significance of these newly detected findings remains unknown at this time. Long-term, longitudinal follow-up of this cohort of patients is needed to determine the natural history and clinical impact of the abnormalities. For patients with both open anterior and posterior arches of C1, instability may develop over time or they may continue to remain asymptomatic. The treating physician may possibly consider a recommendation for certain sports restrictions if these findings were discovered during the course of clinical investigation, although there is no evidence to support a formal recommendation at this time.
Interestingly, several cases of an apparent open posterior arch of C1 on plain radiographs were not verified by advanced imaging. Three cases were found to be closed on CT and five other patients who underwent MRI only also did not have this finding confirmed. The only other instances where advanced imaging was unable to confirm plain radiographic findings occurred with C2–C3 fusions. Two cases of C2–C3 block fusion and two cases of C2–C3 posterior fusion on plain radiographs were not confirmed by MRI, although CT was not performed in these patients. In these instances where only MRI was performed and was unable to confirm a plain radiographic finding, it is unclear whether the finding was not actually present or whether the finding was present but MRI was unable to detect it. For example, C2–C3 fusions in younger patients may not always be evident on MRI because of incomplete ossification. In these instances, fusion needs to be confirmed with flexion and extension radiographs, which demonstrate adjacent vertebral elements moving as a functional unit [13]. In addition, in several cases when both CT and MRI were performed on a patient, only CT was able to detect a particular finding. In four patients with an open anterior arch of C1 that underwent both CT and MRI, only CT demonstrated the abnormality. In two patients with an open posterior arch of C1 that underwent both studies, only CT demonstrated the abnormality. Finally, in two patients with posterior fusion of C2–C3 that underwent both CT and MRI, only CT demonstrated the abnormality. CT may be a more appropriate imaging modality than MRI for detecting bony abnormalities such as these.
The most important benefit provided by advanced imaging in the current study was the ability to evaluate the spinal canal and spinal cord for evidence of canal encroachment and/or cord impingement. Such information may potentially help to define the relationship of encroachment and impingement to other abnormalities detected in the cervical spine and postulate possible neurologic risk. Interestingly, we did not observe any CNS abnormalities in the cervical spine of the 22q11.2 patients, such as syrinx or Chiari type I malformation, as has previously been reported [5, 7, 15]. In addition, only one 22q11.2 patient was found to have neurologic signs and symptoms with corresponding evidence of cervical spinal canal encroachment and spinal cord impingement on advanced imaging. Although most patients were clinically asymptomatic, spinal canal and spinal cord dimensions in the cervical spine were found to be reduced relative to age-matched controls. Significant decreases were noted on both sagittal diameter (mm) and cross sectional area (cm2) measurements on CT and MRI, and included spinal canal sagittal width and axial area, CSF sagittal width and axial area, and spinal cord sagittal width and axial area at one or more cervical levels. Even when not reaching significance, the majority of spinal canal and cord measurements showed smaller mean values in the 22q11.2 patients compared to age-matched controls. These findings were noted even at cervical levels without other bony anomalies, and, typically, were found without a corresponding decrease in the SAC or increase in neurologic symptoms in nearly all of the patients. This suggests that the decreased dimensions of the cervical spine and spinal cord in the 22q11.2 patients represent primary developmental anomalies, rather than secondary effects of the bony anomalies that we have previously described.
Decreased spinal canal and spinal cord dimensions have been suggested as potential markers for present or future neurologic risk in other syndromic patients with cervical spine abnormalities, such as those with Klippel-Feil syndrome [16]. Decreased cross sectional area of the spinal cord has also been shown to correlate with neurologic deficits and pathologic cord changes in other neurologic or spinal disorders, such as multiple sclerosis (MS) [34–43]. It may be hypothesized that the decreased spinal canal and/or spinal cord measurements present in the 22q11.2 population could potentially be predictive of future neurologic risk, particularly as these children grow into adults. However, long-term, longitudinal follow-up of this cohort is needed to determine such an association.
Interestingly, the only cervical spine measurement not found to be significantly decreased relative to age-matched controls in the current study was SAC, which was used to define encroachment (SAC ≤ 13 mm). In retrospect, this definition of encroachment may not be useful in the 22q11.2 patient population, as SAC assesses the sagittal diameter of the spinal canal only at the level of C1. While decreased spinal canal dimensions were clearly seen at the cervical levels below C1 in this study, the majority of patients had an opening of either the anterior or posterior arch of C1, with nearly half (15 patients) having both arches open. The increased space provided by one or both open arches may explain why SAC was not significantly decreased relative to age-matched controls, and why encroachment as defined by SAC was a rare finding for 22q11.2 patients.
In conclusion, we have used advanced imaging to further define the morphology of the cervical spine and spinal cord in patients with the 22q11.2 deletion syndrome. Both CT and MRI were able to detect abnormalities not previously observed on plain radiographs, most notably, evidence of cervical spinal canal encroachment and/or spinal cord impingement in several patients and decreased spinal canal and spinal cord dimensions as a group relative to age-matched controls. However, the clinical significance of these findings is unknown at this time. The decreased dimensions of the spinal canal and spinal cord throughout much of the cervical spine appear to represent primary developmental defects, rather than secondary effects, and, as such, should be considered as significant as other previously described congenital anomalies. Long-term, longitudinal studies following these patients into adulthood are needed to ultimately determine the utility, if any, of these measurements as predictors of neurologic disability.
At present, we recommend routine radiographic evaluation of the cervical spine, including flexion and extension views, in all patients with the 22q11.2 chromosome deletion. Advanced imaging should be pursued in those patients with neurologic signs and/or symptoms suggestive of possible cervical spinal canal encroachment or spinal cord impingement and in those patients with radiographic evidence of cervical spine instability.
Footnotes
Acknowledgments
The authors would like to acknowledge Dr. Allen I. Fleishman, PhD, for the technical assistance that he provided in the statistical analysis.
