Abstract
Importance
Expanded polytetrafluoroethylene (ePTFE) is a widely used alloplast, but studies of augmentation of the nasal tip and nasal dorsum with ePTFE are lacking.
Objective
To investigate whether attaching the conchal cartilage as shield grafts at the distal end of the graft can prevent extrusion and whether the use of ePTFE in the nasal tip can achieve a stable aesthetic outcome.
Design, Setting, and Participants
A randomized clinical trial was performed from April 1, 2006, to October 31, 2008. Follow-up was completed in 2012 and 2017. The study was conducted at the Department of Plastic and Reconstructive Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. One hundred fifty patients seeking rhinoplasty were recruited, and 129 were eligible for inclusion in the study. All the recruited patients were intent-to-treat populations who were randomized into 2 groups: an ePTFE-only group and an ePTFE with conchal cartilage group.
Interventions
Expanded PTFE was used for augmentation of the nasal tip and nasal dorsum in both groups. Conchal cartilage was placed above the distal end in the ePTFE with conchal cartilage group.
Main Outcomes and Measures
Preoperative and postoperative standard photographs were obtained. Postoperative satisfaction and complications were evaluated.
Results
Of the 129 patients (mean age [range], 28 years [21-45 years]; 6 male and 123 female) eligible for inclusion in this study, 76 (mean age [range], 31 years [22-45 years]; 4 male and 72 female) were available for follow-up (mean [range], 106.9 months [100-131 months]) through 2017, including 39 in the ePTFE-only group and 37 in the ePTFE with conchal cartilage group. Infection occurred in 2 patients (5%) in the ePTFE-only group vs 1 patient (3%) in the ePTFE with conchal cartilage group (infection rate [OR, 1.946; 95% CI, 0.169-22.413; P > .99]). Soft-tissue reaction occurred in 0 patients in the ePTFE-only group vs 1 patient (3%) in the ePTFE with conchal cartilage group (soft-tissue reaction rate [OR, 0.000; 95% CI, 0.000-8.538; P = .49]). Irregularity occurred in 0 patients in the ePTFE-only group vs 3 patients (8%) in the ePTFE with conchal cartilage group (irregularity rate [OR, 0.000; 95% CI, 0.000-1.066; P = .11]). Extrusion occurred in neither of the 2 groups.
Conclusions and Relevance
A new technique using ePTFE alone for augmentation of the nasal tip and nasal dorsum achieved safe, attractive, and acceptable outcomes in nasal contouring and patient satisfaction. The use of conchal cartilage as a shield in the nasal tip is not necessary.
Level of Evidence
1.
Trial Registration
Chinese Clinical Trial Registry identifier: ChiCTR-INR-17011101
Key Points
Methods
A prospective randomized clinical trial of patients undergoing rhinoplasty of the nasal tip and nasal dorsum by ePTFE implantation with or without conchal cartilage as a shield graft was performed from April 1, 2006, to October 31, 2008 (Figure 1). The study was conducted at the Department of Plastic and Reconstructive Surgery, Shanghai Ninth People’s Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China. The trial protocol is available in the Supplement. All surgical procedures were performed by one of us (X.L.) using the same technique and material. The study was approved by the institutional review board of the Shanghai Ninth People’s Hospital and adhered to the tenets of the Declaration of Helsinki. Written informed consent was obtained from each participant.
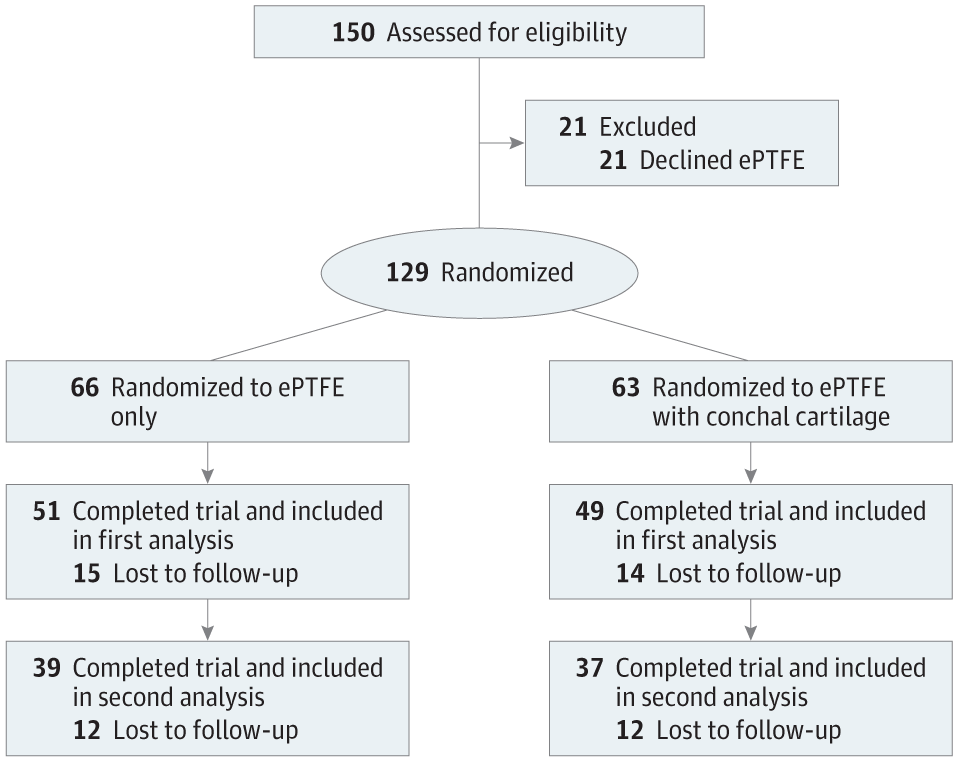
ePTFE indicates expanded polytetrafluoroethylene.
The patients were recruited and then randomized into 2 study groups, an ePTFE-only group and an ePTFE with conchal cartilage group. All the recruited patients were intent-to-treat populations. The method used to generate the random allocation sequence was according to that described by Yu et al.6 Only ePTFE was used in patients assigned odd numbers. Alloplast and conchal cartilage were used in patients assigned even numbers. Preoperative and postoperative standard photographs were obtained. Postoperative satisfaction and complications, including infection, soft-tissue reaction, extrusion, and irregularity, were evaluated. Postoperative follow-up was completed in 2012 and 2017.
Surgical Technique
All procedures were performed with local anesthesia using 1% lidocaine mixed with 1:100 000 epinephrine. Harvesting of conchal cartilage was conducted using a retroauricular approach. We noted the border and avoided damaging the morphologic features of the auricular pavilion. An inverted-V transcolumellar incision with infracartilaginous incisions was made to expose the entire osseocartilaginous framework. The whole nasal skin–soft-tissue pocket was dissected carefully and extensively to reduce the tension on the alloplast and lengthen the nose.
If harvested, conchal cartilage was trimmed into a single-layer shield graft and placed above the caudal border of the lobular part to cover and protect the nasal tip.
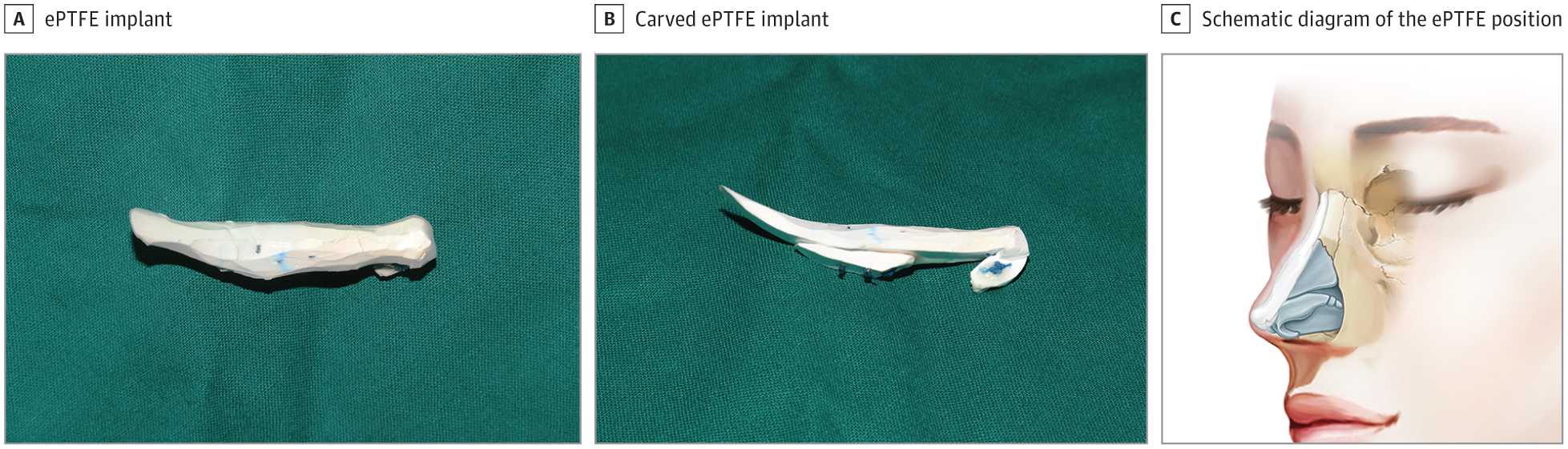
A and B, The L-shaped ePTFE implant was carved to the desired shape. Shown is the same implant after carving with the different viewing angle. C, The schematic diagram was drawn by 2 of us (Y.G. and X.L.) and is published with permission.
Statistical Analysis
The descriptive statistics of the study population were analyzed by frequency for categorical variables and by mean (SD) for normally distributed continuous variables. Occurrence of complications in the 2 groups was compared using Fisher exact test. Two-sided P < .05 was considered to be statistically significant. All analyses were performed using SPSS statistical software, version 19.0 (IBM Corporation).
Results
Of the 129 patients (mean age [range], 28 years [21-45 years]; 6 male and 123 female) recruited into this randomized clinical trial from April 1, 2006, to October 31, 2008, 76 (mean age [range], 31 years [22-45 years]), including 4 men and 72 women, were available for follow-up through March 2017. Thirty-nine patients received rhinoplasty with ePTFE implantation only (ePTFE-only group), and 37 received rhinoplasty with the conchal cartilage as shield grafts above the ePTFE at the nasal tip (ePTFE with conchal cartilage group). A total of 66 patients (87%) had primary cases, and the other 10 patients (13%) had revision cases after an unsuccessful rhinoplasty.
During a mean follow-up of 106.9 months (range, 100-131 months) through 2017, complications associated with ePTFE occurred in 4 of 76 patients (5%), including 3 patients (4%) with infection and 1 patient (1%) with a temporary soft-tissue reaction. Two patients with infection were in the ePTFE-only group, and the remaining 1 patient with infection and 1 patient with soft-tissue reaction were in the ePTFE with conchal cartilage group. The patients with infection did not respond to antibiotic treatment and required implant removal. Infection occurred within 6 months after rhinoplasty in all 4 patients. One primary patient in the ePTFE with conchal cartilage group developed infection within 5 months after rhinoplasty, and a pustule gradually appeared and enlarged on her left nasal tip skin. A revision patient in the ePTFE-only group had poor healing of the right nostril incision within 1 month after rhinoplasty. Pus drained out of the incision when pinching the nasal tip (Figure 3). The remaining patient in the ePTFE with conchal cartilage group also experienced poor healing of the right nostril incision within 1 month after rhinoplasty. The implant was integrally removed through the original incision, and no further augmentation was permitted at the same stage. The patient with soft-tissue reaction had nasal skin redness immediately after surgery that lasted for 4 weeks and then involuted spontaneously.
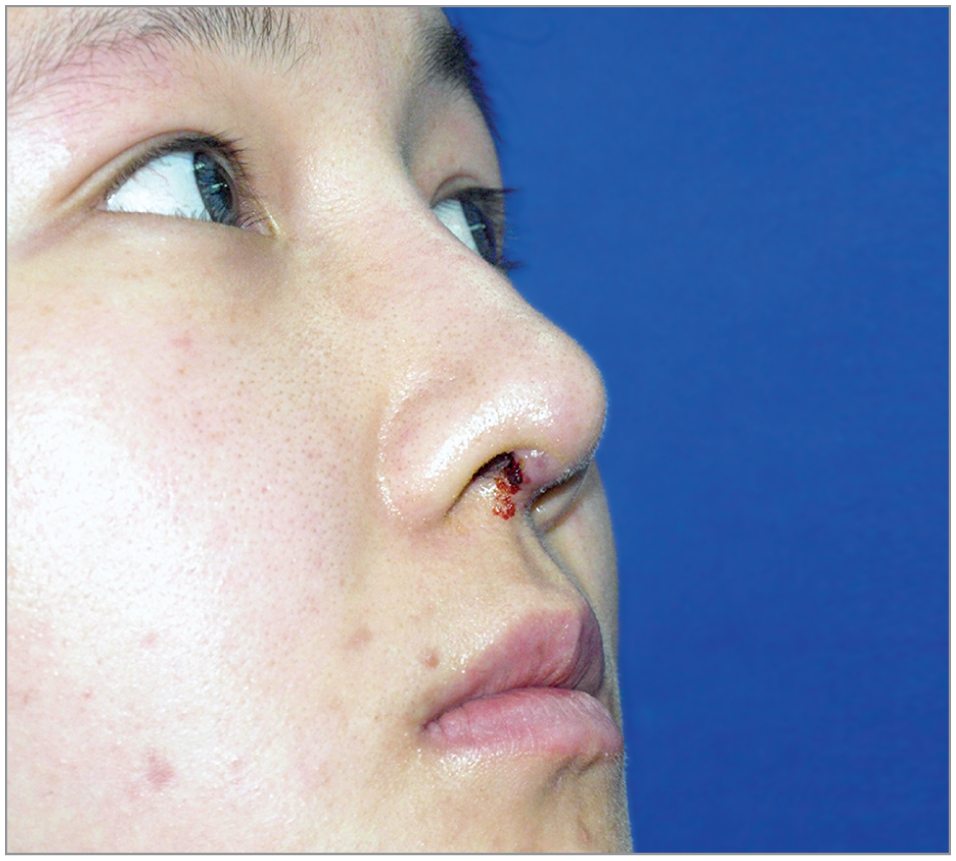
A revision patient in the expanded polytetrafluoroethylene–only group had poor healing of the right nostril incision within 1 month after rhinoplasty. Pus drained out of the incision when pinching the nasal tip.
The following measures were not significantly different between the 2 groups: overall complication rate (odds ratio [OR], 0.346; 95% CI, 0.063-1.906; P = .26), constituent ratio of the complication rate (P = .14), infection rate (OR, 1.946; 95% CI, 0.169-22.413; P > .99), soft-tissue reaction rate (OR, 0.000; 95% CI, 0.000-8.538; P = .49), extrusion rate (OR cannot be calculated; P > .99), and irregularity rate (OR, 0.000; 95% CI, 0.000-1.066; P = .11).
In the ePTFE with conchal cartilage group, 3 of the 37 patients (8%) presented with palpability of the conchal cartilage graft with visible edges of the shield graft and underwent revision; none of the patients in the ePTFE-only group required further correction. There was no significant difference in complication rates between the 2 groups (5.1% in the ePTFE-only group and 13.5% in the ePTFE with conchal cartilage group). No serious displacement was reported in either group. In addition to the 4 patients with infection and soft-tissue reaction, 4 other patients without infection were dissatisfied with their outcome. Two of these 4 patients requested implant removal. The remaining patients in the study obtained satisfactory results. At postoperative clinic visits, no problems were reported related to the position or texture of the implants. During clinical examinations, the alloplast appeared to be well integrated without any contour irregularities. We could not distinguish between the 2 groups based on the tip contour and softness by palpability. The nasal dorsum was significantly augmented after surgery. Enhanced tip projection and downward rotation of the nasal tip were achieved (Figure 4). All nasal incisions healed well without obvious scar. The donor sites showed no disfigurement.
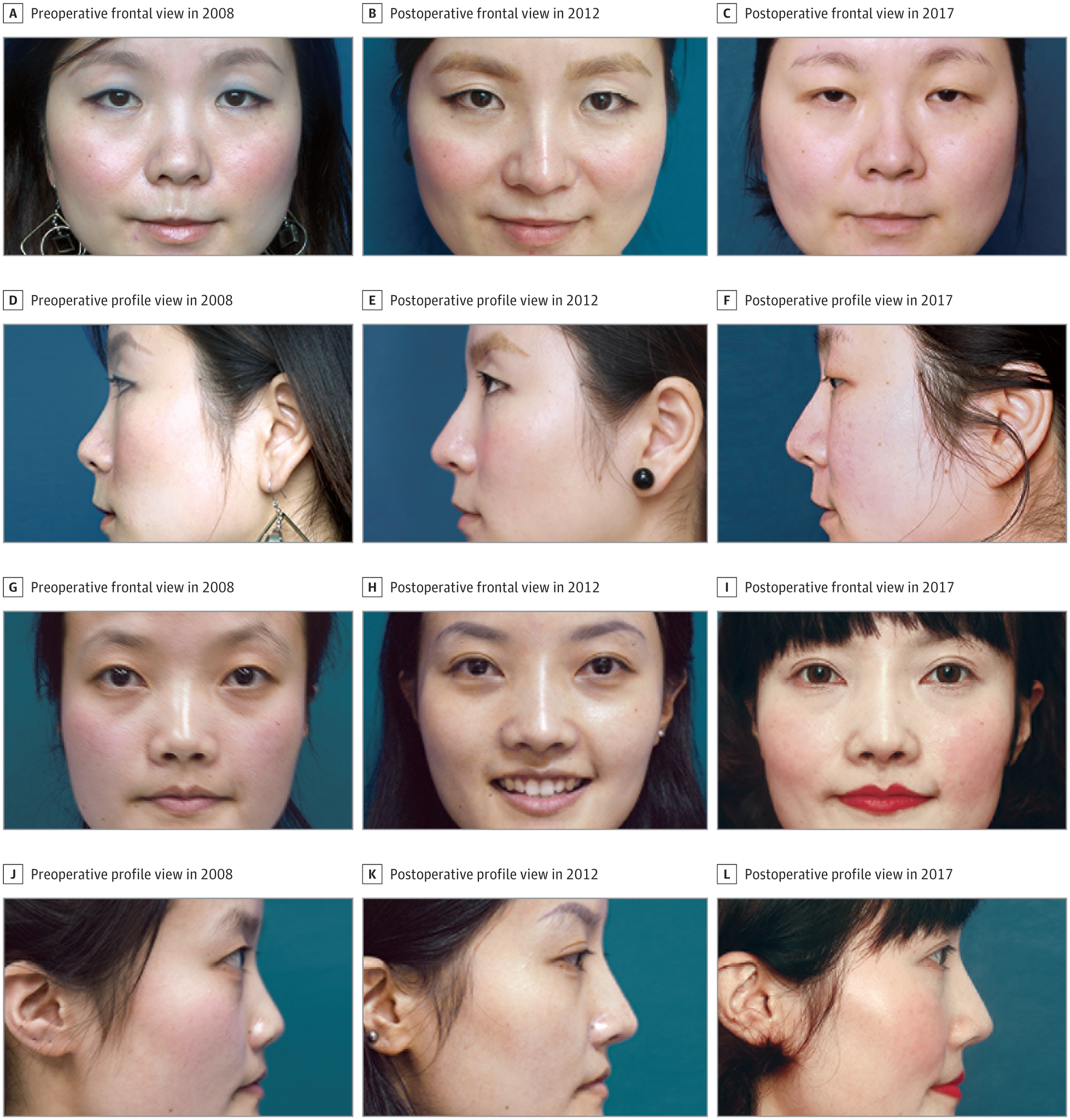
A-F, A patient in the expanded polytetrafluoroethylene (ePTFE)–only group. Compared with the preoperative anthropometric measurements, the postoperative nasal tip projection ratio increased from 0.67 to 0.73, the nasolabial angle decreased from 127.94° to 108.76°, and the columellar facial angle decreased from 118.54° to 113.94°. G-L, A patient in the ePTFE with conchal cartilage group. Compared with the preoperative anthropometric measurements, the postoperative nasal tip projection ratio increased from 0.55 to 0.64, the nasolabial angle decreased from 109.54° to 102.16°, and the columellar facial angle decreased from 117.30° to 111.89°.
Discussion
Widely known as Gore-Tex (W. L. Gore Associates) in the West, ePTFE has been used clinically since it was introduced as a vascular prosthesis.8 Owing to the long-term efficacy, safety, and biocompatibility established in vascular surgery,9,10 ePTFE has been used extensively for abdominal wall defect, hernia, and facial reconstruction,11 as well as other indications. The material was first described in rhinoplasty augmentation by Rothstein and Jacobs12 in 1989 and was subsequently approved in 1993 by the US Food and Drug Administration for use in rhinoplasty and other aesthetic surgical procedures. Expanded PTFE is a polymer of carbon bound to fluorine formed into an inert weave of PTFE nodules and thin PTFE fibrils.13 It is a microporous alloplastic compound, with pore sizes ranging from 10 to 30 μm (mean, 22 μm), that provides sufficient tissue ingrowth and fixation of the implant. Several animal models and clinical experience have confirmed the high biocompatibility and safety of ePTFE.14-17 Minimal chronic inflammation and foreign-body reaction were observed in those studies. A tiny fibrotic capsule forms around the implant.14-16 For our routine operations, we selected Tisuthes (Shanghai Suokang Medical Implants), a widely used ePTFE implant brand in China that can provide different models of shape and hardness.
To our knowledge, the present study is the first specialized, prospective randomized clinical trial to use ePTFE in augmentation of the nasal tip and nasal dorsum. Before this trial, we had accumulated years of experience using ePTFE alone to augment the nasal tip and nasal dorsum. Previous studies7,18-20 mainly focused on the safety and efficacy of using ePTFE in the nasal dorsum only; the infection rates in the previous studies ranged from 0% to 3.2%. These results are similar to those obtained in the present study. Insufficient evidence was found to conclude that the application of ePTFE in the nasal tip may increase the infection rate during long-term follow-up.
Moreover, no patient in either group experienced alloplast extrusion during the 10-year follow-up.
In the early stage of our prospective randomized clinical trial, 3 patients had palpability of the conchal cartilage graft with visible edges of a shield graft in the nasal tip and required revision. No similar complications occurred after increased attention was given to the surgical techniques of creating smooth and round corners at both the alloplast and the shield cartilage. We concluded that localized dysvascularity26 or hypertension after surgery caused the soft-tissue reaction that manifested skin redness of the nasal tip in 1 patient and considered that the spontaneous improvement 4 weeks later might have resulted from the revascularization of the damaged tissue or adaptation to high pressure.
As previously mentioned, a weak and small cartilaginous framework, in addition to thick skin, requires a rigid and strong structural support, which necessitates harvesting costal cartilage. Dorsal augmentation using diced conchal cartilage is limited by its shortcomings of material, inclusion resorption, and long duration of the procedure.27,28 Costal cartilage provides a sufficient supply of graft material necessary not only for structural support to allow tip rotation but also for dorsal augmentation.29 Concerns regarding additional obvious chest scarring that the inframammary fold may fail to hide discourage the use of costal cartilage among Asian patients. In addition to donor site morbidity, patients are also concerned about rigidity, warping, and resorption of the costal cartilage. One study30 concluded that dorsal augmentation with an autologous costal cartilage leads to similar aesthetic outcomes but with a higher complication rate compared with dorsal augmentation with ePTFE.
The septal extension graft is an effective technique in rhinoplasty because it can reshape the nasal tip; the septal cartilage is the most commonly used material. However, the septal cartilage is considerably weaker, thinner, and smaller in Asian patients, particularly those with short-nose deformities.31,32
Compared with other techniques in rhinoplasty, the use of ePTFE in the nasal tip and nasal dorsum requires less operative time (usually <40 minutes). Given our experience, we generally carve the alloplast once or perform a slight revision depending on the nasal contour. It is inconclusive whether operative time is an independent risk factor for complications; some surgeons report that the complication rate increases significantly after 3 hours.33-35 Short operative time and minimal trauma of surgery improve patient comfort and allow for quick recovery. On the basis of our experience, ePTFE can be removed integrally and be replaced with autogenous cartilage grafts in a second stage if patients develop infection or are dissatisfied with the material.
We believe that the characteristics of the ePTFE material and the Asian nose contribute to the safety of the implant. Tissue incorporation, cell attachment, and capsule formation reduce the tendency of the ePTFE to deteriorate. Thicker sebaceous skin also acts as a strong wall to decrease tension over the implant36 and prevent extrusion.
Limitations
The major limitation of our prospective randomized clinical trial is the withdrawal bias attributed to a decade of follow-up. This factor may have affected the complication rate. Another limitation is that no patient-reported outcome measure tool was used to assess patient satisfaction.
Conclusions
A new technique using ePTFE alone for augmentation of the nasal tip and nasal dorsum achieved safe and attractive outcomes in nasal contouring and obtained satisfactory patient results. The use of conchal cartilage as a shield in the nasal tip is not necessary.
